

Cyclobenzaprine
- Molecular FormulaC20H21N
- Average mass275.387 Da
- MK-130
- TNX-102
1-(3-Dimethylaminopropylidene)-2,3:6,7-dibenzo-4-suberene
1-Propanamine, 3-(5H-dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-[ACD/Index Name]
206-145-8[EINECS]
3-(5H-Dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-1-propanamine
303-53-7[RN]
5-(3-Dimethylaminopropylidene)dibenzo[a,e]cycloheptatriene
циклобензаприн[Russian][INN]
سيكلوبنزابرين[Arabic][INN]
环苯扎林[Chinese][INN]
Cyclobenzaprine, CAS Registry Number: 303-53-7
CAS Name: 3-(5H-Dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-1-propanamine
Additional Names:N,N-dimethyl-5H-dibenzo[a,d]cyclohepten-D5,g-propylamine; 5-(3-dimethylaminopropylidene)dibenzo[a,e]cycloheptatriene; 1-(3-dimethylaminopropylidene)-2,3:6,7-dibenzo-4-suberene; proheptatriene
Manufacturers’ Codes: MK-130; Ro-4-1577; RP-9715
Molecular Formula: C20H21N, Molecular Weight: 275.39
Percent Composition: C 87.23%, H 7.69%, N 5.09%
Literature References: Prepn: GB858187 (1961 to Hoffmann-La Roche); Villani et al.,J. Med. Pharm. Chem.5, 373 (1962); Winthrop et al.,J. Org. Chem.27, 230 (1962). Pharmacology: C. D. Barnes, W. L. Adams, Neuropharmacology17, 445 (1978); N. N. Share, ibid. 721; and toxicology: J. Metysova et al.,Arch. Int. Pharmacodyn. Ther.144, 481 (1963). Metabolism: G. Belvedere et al.,Biomed. Mass Spectrom.1, 329 (1974); H. B. Hucker et al.,Drug Metab. Dispos.6, 184 (1978). Bioavailability: eidem,J. Clin. Pharmacol.17, 719 (1977). Clinical studies: J. V. Basmajian, Arch. Phys. Med. Rehabil.5, 58 (1978); B. R. Brown, J. Womble, J. Am. Med. Assoc.240, 1151 (1978). Comprehensive description: M. L. Cotton, G. R. B. Down, Anal. Profiles Drug Subs.17, 41-72 (1988).
Properties: bp1 175-180°. uv max: 224, 289 nm (log e 4.57, 4.02), (Villani et al.)
Boiling point: bp1 175-180°
Absorption maximum: uv max: 224, 289 nm (log e 4.57, 4.02), (Villani et al.)
Derivative Type: Hydrochloride
CAS Registry Number: 6202-23-9
Trademarks: Flexeril (Merck & Co.); Flexiban (Merck & Co.)
Molecular Formula: C20H21N.HCl, Molecular Weight: 311.85
Percent Composition: C 77.03%, H 7.11%, N 4.49%, Cl 11.37%
Literature References: Use as muscle relaxant: N. N. Share, FR2100873 (1972 to Frosst), C.A.78, 47801n (1973).
Properties: Crystals from isopropanol, mp 216-218°. Soly in water: >20 g/100 ml. Freely sol in water, methanol, ethanol; sparingly sol in isopropanol; slightly sol in chloroform, methylene chloride. Practically insol in hydrocarbons. uv max: 226, 295 nm (e 52300, 12000). LD50 in mice (mg/kg): 35 i.v., 250 orally (Metysova).
Melting point: mp 216-218°
Absorption maximum: uv max: 226, 295 nm (e 52300, 12000)
Toxicity data: LD50 in mice (mg/kg): 35 i.v., 250 orally (Metysova)
Therap-Cat: Muscle relaxant (skeletal).
Keywords: Muscle Relaxant (Skeletal).
Cyclobenzaprine, a centrally-acting muscle relaxant, was first synthesized in 196111 and has been available for human use since 1977.10 It was initially studied for use as antidepressant given its structural similarity to tricyclic antidepressants – it differs from Amitriptyline by only a single double bond.11,10 Since its approval, it has remained relatively popular as an adjunctive, short-term treatment for acute skeletal muscle spasms secondary to musculoskeletal injury.
Cyclobenzaprine (sold under the brand name Flexeril, among others) is a medication used for muscle spasms from musculoskeletal conditions of sudden onset.[6] It is not useful in cerebral palsy.[6] It is taken by mouth.[6] Use is not recommended for more than a few weeks.[6]
Common side effects include headache, feeling tired, dizziness, and dry mouth.[6] Serious side effects may include an irregular heartbeat.[6] There is no evidence of harm in pregnancy, but it has not been well studied in this population.[6] It should not be used with an MAO inhibitor.[6] How it works is unclear.[6]
Cyclobenzaprine was approved for medical use in the United States in 1977.[6] It is available as a generic medication.[6] In 2019, it was the 45th most commonly prescribed medication in the United States, with more than 15 million prescriptions.[7][8] It was not available in the United Kingdom as of 2012.[9]
Synthesis Reference
Villani, F.J.; US. Patent 3,409,640; November 5,1968; assigned to Schering Corporation.
Paper
By: Gowda, Narendra B.; Rao, Gopal Krishna; Ramakrishna, Ramesha A.
Tetrahedron Letters (2010), 51, (43), 5690-5693.
https://www.sciencedirect.com/science/article/abs/pii/S0040403910014668
A simple and convenient protocol for deoxygenation of aliphatic and aromatic N-oxides to the corresponding amines in good to excellent yield using sodium borohydride–Raney nickel in water is reported. Other functional moieties such as alkenes, halides, ethers, and amides are unaffected under the present reaction condition.
Graphical abstract

Cyclobenzaprine N-oxide, CAS RN: 6682-26-4
Dissolve (1 mmol) of cyclobenzaprine N-oxide in 2.5 mL of water at 60 °C. 2. Add Raney nickel (0.10 g, W6 grade) to the solution. 3. Stir the reaction mixture for 10 minutes. 4. Add (2 mmol) of sodium borohydride slowly in portions over 15-20 minutes to the reaction mixture. 5. Stir the reaction mixture at the same temperature for 2.5 hours (the completion of the reaction as monitored by TLC). 6. Once the reaction is completed, add chloroform (50 mL) to the reaction mixture. 7. Filter the resulted mixture to remove Raney nickel. 8. Dry the chloroform layer over anhydrous magnesium sulfate. 9. Filter the reaction mixture. 10. Evaporate the solvent under vacuum. 11. Purify the obtained residue through short path flash chromatography with silica gel and chloroform.
1H NMR (400 MHz, CDCl3) δ: 1.12 (s, 6H, N-CH3), 1.23- 1.34 (m, 4H, CH2), 4.58 (t, J= 4.0 Hz, 1H, CH), 5.82(d, J= 4.0 Hz, 2H, CH), 6.21- 6.33 (m, 8H, ArH).
13C NMR (100 MHz, CDCl3) δ: 27.89, 45.93, 60.12, 127.40, 127.55, 128.30, 128.59, 128.92, 129.33, 129.45, 129.67, 131.74, 131.96, 132.40, 134.63, 135.39, 137.97, 142.95, 143.30.
SYN
PATENT
https://patents.google.com/patent/WO2012098563A2/en
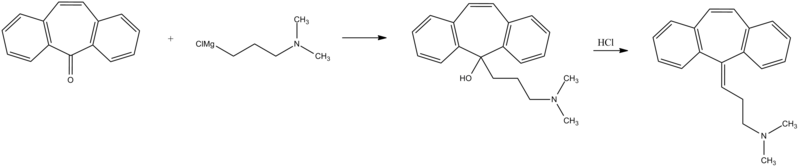
Cyclobenzaprine hydrochloride, chemically known as 5-(3-dimethylaminopropylidene)- dibenzo (a,e) cycloheptatriene hydrochloride (Formula I),

Formula I is a commonly prescribed tricyclic amine having muscle relaxant pharmaceutical activity. After sustaining an injury, muscle spasms may occur to stabilize the affected body part and prevent further damage. Cyclobenzaprine hydrochloride is used to treat such muscle spasm associated with acute, painful musculoskeletal conditions.
Few multistep processes for the preparation of this tricyclic amine are already available in the literature which involves isolation and purification of intermediate compounds. The conventional route of synthesis as reported in US3454643, ES8201950 includes preparation of Grignard reagent (GR) of 3-dimethylaminopropyl chloride in a first step, reacting with 5-dibenzosuberenone (Formulall) in a second step. The reaction mass was extracted with benzene, solid obtained was recrystallized from alcohol to produce 5- hydroxy intermediate (Formula III) and further dehydrated in third step using acetyl chloride or acetic anhydride in presence of chloroform as a solvent medium followed by purging HC1 gas to produce hydrochloride salt (Formula I). CH,
CI-(CH2)3 NS
CH,
Dimeth laminopropyl chloide

Di methy lam i nopropy I 5-dibenzosubrenone – y roxy compoun magnesium chloide
(Formula II) (Formula III)

Cyclobenzaprine base Cyclobenzaprine hydrochloride
(Formula IV) (Formula I)
The multistep synthesis is cumbersome and use of hazardous solvents and reagents like chloroform, benzene and acetyl chloride etc are not recommended for the preparation of pharmaceutical substances.
J. Org. Chem. Vol. 27, 230-240 (1961) also portrayed similar procedure for the synthesis of cyclobenzaprine hydrochloride, wherein 5-hydroxy compound of formula III was isolated and recrystallized before dehydration reaction.
Synthetic Comm. 11 (3), 241-246 (1981) described a process which involves isolation and purification of the intermediate at magnesium -complex stage. Hydrolysis of the isolated complex afforded desired tricyclic amine. GB858186 and GB858187 jointly described a process which comprises preparation of 5- hydroxy compound (Formula III) and subsequent conversion of the same to cyclobenzaprine hydrochloride. However the overall yield reported is significantly low.
In a different approach, a high temperature dehydrogenation of amitriptyline base resulting in formation of cyclobenzaprine hydrochloride is reported in Indian patent application 387/CHE/2005.

. EXAMPLE:
In a reaction vessel, THF (1 10ml), magnesium turnings 20gm (0.823mole) were charged and the mixture was warmed to 45-55°C for 20 min. A solution of l OOgm (0.823mole) of 3-dimethylaminopropyl chloride prepared in 1 10ml THF was added dropwise to the reaction mixture by controlling the reflux generated due to reaction initiation and maintained for 2hrs. The formed Grignard reagent was then cooled to 0-5°C and a solution of lOOgm (0.485mole) 5-dibenzosuberenone prepared in 220ml THF was charged to the reaction mass at temperature below 10°C. The reaction mass was stirred for 45 min at temperature 10-15°C. The absence of 5-dibenzosuberenone was checked by TLC and 770ml of 20% aq. HC1 was charged to the reaction mass at a temperature below 10°C. The reaction mass was then heated to 70-80°C for 3 hrs. The acidic mass was neutralized by using aqueous Na2C03 solution and extracted with 900ml methylene dichloride. The solvent was removed completely under reduced pressure and oil thus formed was dissolved in 450ml IPA and acidified with 240 ml of 20% IPA .HC1 solution and stirred for 2 hrs at 0-5°C for complete precipitation. The precipitate is filtered, recrystallized from IPA (800 ml) and dried to obtain 1 18 gm (78%) white crystalline cyclobenzaprine hydrochloride with purity 99.93% by HPLC.

PATENT
PATENT
CN 111393305
CLIP
Muscle Relaxants
R.S. Vardanyan, V.J. Hruby, in Synthesis of Essential Drugs, 2006
Cyclobenzaprine
Cyclobenzaprine, N,N-dimethyl-3-(dibenzo[a,d]cyclohepten-5-ylidene) propylamine (15.3.9), is synthesized by reacting 5H-dibenzo[a,d]cyclohepten-5-one with 3-dimethylaminopropylmagnesium chloride and subsequent dehydration of the resulting carbinol (15.3.8) in acidic conditions into cyclobenzaprine (15.3.9) [30–32].

Cyclobenzaprine is structurally similar to tricyclic antidepressants. It acts at the brain stem level. It is used as an adjuvant agent for relieving muscle spasms associated with severe diseased conditions of the muscle. A synonym of this drug is flexeril.
///////////////////////////////////////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Medical use
Cyclobenzaprine is used, in conjunction with physical therapy, to treat muscle spasms that occur because of acute musculoskeletal conditions.[10] After sustaining an injury, muscle spasms to stabilize the affected body part occur, which may increase pain to prevent further damage. Cyclobenzaprine is used to treat such muscle spasms associated with acute, painful musculoskeletal conditions.[11] It decreases pain in the first two weeks,[12][13] peaking in the first few days, but has no proven benefit after two weeks.[12][14] Since no benefit is proven beyond that, therapy should not be continued long-term.[11] It is the best-studied muscle relaxer.[12] It is not useful for spasticity due to neurologic conditions such as cerebral palsy.[11][15]
A 2004 review found benefit for fibromyalgia symptoms, with a reported number needed to treat of 4.8 (meaning that 1 person out of every 4.8 benefits from treatment) for pain reduction, but no change in fatigue or tender points.[16] A 2009 Cochrane review found insufficient evidence to justify its use in myofascial pain syndrome.[17] It may also be used along with other treatments for tetanus.[18]
Side effects
Cyclobenzaprine results in increased rates of drowsiness (38%), dry mouth (24%), and dizziness (10%).[14] Drowsiness and dry mouth appear to intensify with increasing dose.[19] The sedative effects of cyclobenzaprine are likely due to its antagonistic effect on histamine, serotonin, and muscarinic receptors.[medical citation needed]
Agitation is a common side effect observed, especially in the elderly. Some experts[who?] believe that cyclobenzaprine should be avoided in elderly patients because it can cause confusion, delirium, and cognitive impairment.[20][21] In general, the National Committee for Quality Assurance recommends avoiding the use of cyclobenzaprine in the elderly because of the potential for more severe side effects.[22]
Dysphagia, a life-threatening side-effect, may rarely occur.[23] Treatment protocols and support should follow the same as for any structurally related tricyclic, such as tricyclic antidepressants.[24]
Overdose
The most common effects of overdose are drowsiness and tachycardia.[11] Rare but potentially critical complications are cardiac arrest, abnormal heart rhythms, severe low blood pressure, seizures, and neuroleptic malignant syndrome.[11] Life-threatening overdose is rare,[11] however, as the median lethal dose is about 338 milligrams/kilogram in mice and 425 mg/kg in rats.[11] The potential harm is increased when central nervous system depressants and antidepressants are also used; deliberate overdose often includes alcohol among other drugs.[11]
Interactions
Cyclobenzaprine has major contraindications with monoamine oxidase inhibitors (MAOIs). At least one study also found increased risk of serotonin syndrome when cyclobenzaprine was taken with the serotonergic drugs duloxetine or phenelzine.[25]
These substances may interact with cyclobenzaprine:
- Central nervous system depressants (e.g. alcohol, opioids, benzodiazepines, nonbenzodiazepines, phenothiazines, carbamates, barbiturates, major tranquilizers)
- Monoamine oxidase inhibitors taken within two weeks of cyclobenzaprine may result in serious, life-threatening side effects.[11]
Cyclobenzaprine may affect the medications used in surgical sedation and some surgeons request that patients temporarily discontinue its use prior to surgery.[26]
Pharmacology
Cyclobenzaprine is a centrally acting muscle relaxant.[27] Cyclobenzaprine is a 5-HT2 receptor antagonist; it relieves muscle spasm through action on the central nervous system at the brain stem, rather than targeting the peripheral nervous system or muscles themselves.[28]
Pharmacodynamics
| Site | CBP | NCBP | Action | Ref |
|---|---|---|---|---|
| 5-HT1A | 5.3 | 3.2 | Agonist | [29] |
| 5-HT2A | 5.2 | 13 | Antagonist | [29] |
| 5-HT2B | 100 | ??? | Antagonist | [29] |
| 5-HT2C | 5.2 | 43 | Antagonist | [29] |
| α1A | 5.6 | 34 | ND | [29] |
| α2A | 4.3 | 6.4 | Antagonist | [29] |
| α2B | 21 | 150 | ND | [29] |
| α2C | 21 | 48 | ND | [29] |
| H1 | 1.3 | 5.6 | ND | [29] |
| M1 | 7.9 | 30 | ND | [29] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. |
Pharmacokinetics
Cyclobenzaprine has an oral bioavailability of about 55% and approximately 93% is bound to proteins in plasma. The half-life of the drug is 18 hours and it has a plasma clearance of 0.7 litres per minute.[27][30][31]
Comparison to other medications
Cyclobenzaprine has been found to be not inferior to tizanidine, orphenadrine, and carisoprodol in the treatment of acute lower back pain, although none have been proven to be effective for long-term use (beyond two weeks of treatment). No differences in pain or spasm scores were noted among these agents, nor when compared to benzodiazepines.[32] However, nonbenzodiazepine (including cyclobenzaprine) treatment was found to have a lower risk of medication abuse and continuation of use against medical advice.[medical citation needed] Side effects such as sedation and ataxia are also less pronounced with nonbenzodiazepine antispasmodics.[medical citation needed]
In a study on the treatment of musculoskeletal pain treatment with cyclobenzaprine alone or in combination with ibuprofen, no significant differences in pain scores were noted among the three treatment groups. Peak benefit was found to occur on day seven of the treatment for all groups.[33]
Formulations
Cyclobenzaprine 10mg tablets
By mouth, cyclobenzaprine is marketed as Apo-Cyclobenzaprin, Fexmid, Flexeril and Novo-Cycloprine. It is available in generic form. A once-a-day, extended-release formulation, Amrix, is available.[34] Cyclobenzaprine is also used by compounding pharmacies in topical creams.[citation needed]
References
- ^ Micromedex® 2010 – DRUGDEX Evaluations (Cyclobenzaprine Hydrochloride)
- ^ “Cyclobenzaprine Hydrochloride Tablets USP Revised: April 2005 Rx only”. nih.gov. Retrieved 1 October 2016.
- ^ Teva Pharmaceuticals USA, Inc (May 2016). “AMR40470 (Amrix) Prescribing Information” (PDF).
- ^ U.S. Food and Drug Administration. “NDA 17-821/S-045 Flexeril (Cyclobenzaprine HCl) Tablets” (PDF).
- ^ Teva Pharmaceuticals USA, Inc (May 2016). “AMR40470 (Amrix) Prescribing Information” (PDF).
- ^ Jump up to:a b c d e f g h i j k “Cyclobenzaprine Monograph for Professionals”. Drugs.com. AHFS. Retrieved 22 December 2018.
- ^ “The Top 300 of 2019”. ClinCalc. Retrieved 16 October 2021.
- ^ “Cyclobenzaprine – Drug Usage Statistics”. ClinCalc. Retrieved 16 October 2021.
- ^ “Fibromyalgia, psychiatric comorbidity, and the somatosensory cortex”. British Journal of Medical Practitioners. 5 (2): a522. 2012.
- ^ Yang YW, Macdonald JB, Nelson SA, Sekulic A (December 2017). “Treatment of vismodegib-associated muscle cramps with cyclobenzaprine: A retrospective review”. Journal of the American Academy of Dermatology. 77 (6): 1170–1172. doi:10.1016/j.jaad.2016.12.017. PMID 29132849. S2CID 8265576.
- ^ Jump up to:a b c d e f g h i “Cyclobenzaprine- cyclobenzaprine hydrochloride tablet, film coated”. DailyMed. 30 December 2019. Retrieved 26 September 2020.
- ^ Jump up to:a b c Chou R, Peterson K, Helfand M (August 2004). “Comparative efficacy and safety of skeletal muscle relaxants for spasticity and musculoskeletal conditions: a systematic review”. Journal of Pain and Symptom Management. 28 (2): 140–75. doi:10.1016/j.jpainsymman.2004.05.002. PMID 15276195.
- ^ van Tulder MW, Touray T, Furlan AD, Solway S, Bouter LM (2003). Van Tulder MW (ed.). “Muscle relaxants for non-specific low back pain”. The Cochrane Database of Systematic Reviews. 2 (2): CD004252. doi:10.1002/14651858.CD004252. PMC 6464310. PMID 12804507.
- ^ Jump up to:a b Browning R, Jackson JL, O’Malley PG (July 2001). “Cyclobenzaprine and back pain: a meta-analysis”. Archives of Internal Medicine. 161 (13): 1613–20. doi:10.1001/archinte.161.13.1613. PMID 11434793.
- ^ Ashby P, Burke D, Rao S, Jones RF (October 1972). “Assessment of cyclobenzaprine in the treatment of spasticity”. Journal of Neurology, Neurosurgery, and Psychiatry. 35 (5): 599–605. doi:10.1136/jnnp.35.5.599. PMC 494138. PMID 4563483.
- ^ Tofferi JK, Jackson JL, O’Malley PG (February 2004). “Treatment of fibromyalgia with cyclobenzaprine: A meta-analysis”. Arthritis and Rheumatism. 51 (1): 9–13. doi:10.1002/art.20076. PMID 14872449.
- ^ Leite FM, Atallah AN, El Dib R, Grossmann E, Januzzi E, Andriolo RB, da Silva EM (July 2009). “Cyclobenzaprine for the treatment of myofascial pain in adults”. The Cochrane Database of Systematic Reviews (3): CD006830. doi:10.1002/14651858.CD006830.pub3. PMC 6481902. PMID 19588406.
- ^ Smith BT (2014). Pharmacology for Nurses. Jones & Bartlett Publishers. p. 122. ISBN 9781449689407.
- ^ “Flexeril: Side effects”. RxList.com. Archived from the original on 12 September 2008. Retrieved 22 February 2010.
- ^ “Long-term Use of Cyclobenzaprine for Pain: A Review of the Clinical Effectiveness”. CADTH Rapid Response Reports. Ottawa, Ontario: Canadian Agency for Drugs and Technologies in Health. 23 February 2015. PMID 25763449.
- ^ Potentially inappropriate medications for the elderly according to the revised Beers criteria. 2012. Duke Clinical Research Institute website. [1]
- ^ “High risk medications” (PDF). National Committee for Quality Assurance. Archived from the original (PDF) on 1 February 2010. Retrieved 22 February 2010.
- ^ “MEDICATIONS AND DYSPHAGIA/ SWALLOWING RISKS” (PDF).
- ^ Chabria SB (July 2006). “Rhabdomyolysis: a manifestation of cyclobenzaprine toxicity”. Journal of Occupational Medicine and Toxicology. 1 (1): 16. doi:10.1186/1745-6673-1-16. PMC 1540431. PMID 16846511.
- ^ Keegan MT, Brown DR, Rabinstein AA (December 2006). “Serotonin syndrome from the interaction of cyclobenzaprine with other serotoninergic drugs”. Anesthesia and Analgesia. 103 (6): 1466–8. doi:10.1213/01.ane.0000247699.81580.eb. PMID 17122225.
- ^ Medical Practice of William H. Gorman, M.D. (18 February 2014). “Medications to Avoid, Continue, or Stop – Before & After Surgery”.
- ^ Jump up to:a b “Cyclobenzaprine”. http://www.drugbank.ca.
- ^ Kobayashi H, Hasegawa Y, Ono H (September 1996). “Cyclobenzaprine, a centrally acting muscle relaxant, acts on descending serotonergic systems”. European Journal of Pharmacology. 311 (1): 29–35. doi:10.1016/0014-2999(96)00402-5. PMID 8884233.
- ^ Jump up to:a b c d e f g h i j k “Cyclobenzaprine (CBP) and Its Major Metabolite Norcyclobenzaprine (nCBP) Are Potent Antagonists of Human Serotonin Receptor 2a (5HT2a), Histamine Receptor H-1 and á-Adrenergic Receptors: Mechanistic and Safety Implications for Treating Fibromyalgia Syndrome by Improving Sleep Quality”. ACR Meeting Abstracts. Retrieved 27 January 2022.
- ^ “Cyclobenzaprine”. pubchem.ncbi.nlm.nih.gov.
- ^ Winchell GA, King JD, Chavez-Eng CM, Constanzer ML, Korn SH (January 2002). “Cyclobenzaprine pharmacokinetics, including the effects of age, gender, and hepatic insufficiency”. Journal of Clinical Pharmacology. 42 (1): 61–9. doi:10.1177/0091270002042001007. PMID 11808825. S2CID 7749001.
- ^ “Medscape: Medscape Access”. medscape.com. Retrieved 1 October 2016.
- ^ Childers MK, Petri M, Laudadio C, Harrison D, Silber S, Bowen D (2004). “Comparison of cyclobenzaprine alone versus cyclobenzaprine plus ibuprofen in patients with acute musculoskeletal spasm and pain”. Annals of Emergency Medicine. 44 (4): S87–S88. doi:10.1016/j.annemergmed.2004.07.286.
- ^ “Patient Web site for Amrix (Cyclobenzaprine Hydrochloride Extended‐Release Capsules)”. amrix.com. Retrieved 1 October 2016.
External links
- “Cyclobenzaprine”. Drug Information Portal. U.S. National Library of Medicine.
| Clinical data | |
|---|---|
| Trade names | Flexeril, Amrix, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682514 |
| License data | US DailyMed: Cyclobenzaprine |
| Routes of administration | By mouth |
| ATC code | M03BX08 (WHO) |
| Legal status | |
| Legal status | US: ℞-onlyIn general: ℞ (Prescription only) |
| Pharmacokinetic data | |
| Bioavailability | 33–55%[1][2] |
| Protein binding | 93% |
| Metabolism | major: CYP3A4, CYP1A2; minor: CYP2D6, N-demethylation[5] |
| Metabolites | Norcyclobenzaprine |
| Elimination half-life | 32 hours (extended-release, range 8-37 hours),[3] 18 hours (immediate release, range 8–37 hours)[4] |
| Excretion | Kidney |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 303-53-7 |
| PubChem CID | 2895 |
| IUPHAR/BPS | 7152 |
| DrugBank | DB00924 |
| ChemSpider | 2792 |
| UNII | 69O5WQQ5TI |
| KEGG | D07758 |
| ChEBI | CHEBI:3996 |
| ChEMBL | ChEMBL669 |
| CompTox Dashboard (EPA) | DTXSID0046933 |
| ECHA InfoCard | 100.005.588 |
| Chemical and physical data | |
| Formula | C20H21N |
| Molar mass | 275.395 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (verify) |
///////////////cyclobenzaprine, циклобензаприн , سيكلوبنزابرين , 环苯扎林 , MK-130, TNX-102, Muscle Relaxant
CN(C)CCC=C1C2=CC=CC=C2C=CC2=CC=CC=C12
