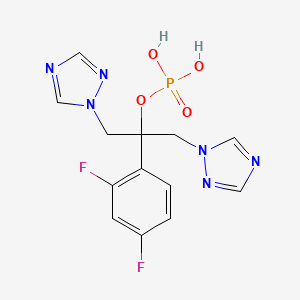
Fosfluconazole
Fosfluconazole; 194798-83-9; UNII-3JIJ299EWH; 3JIJ299EWH; NCGC00182029-01;
2-(2,4-difluorophenyl)-1,3-di(1h-1,2,4-triazol-1-yl)propan-2-yl dihydrogen phosphate;
2,4-difluoro-α,α-bis(1H-1,2,4-triazol-1-ylmethyl) benzyl alcohol, dihydrogen phosphate
| Molecular Formula: | C13H13F2N6O4P |
|---|---|
| Molecular Weight: | 386.250688 g/mol |
Research Code:UK-292663, UK 292663, F-FLCZ, F FLCZ
Trade Name:Prodif® PFIZER
MOA:Azole antifungal
Indication:Cryptococcus neoformans; Candidiasis
Status:Approved, Japan PMDA OCT 16 2003
Company:Pfizer (Originator)
Candidiasis,Cryptococcus neoformans, Injection, Solution, Eq. 100 mg/200 mg/400 mg fluconazole per vial
Fosfluconazole (INN) is a water-soluble phosphate prodrug of fluconazole – a triazole antifungal drug used in the treatment and prevention of superficial and systemic fungal infections. The phosphate ester bond is hydrolysed by the action of a phosphatase – an enzyme that removes a phosphate group from its substrate by hydrolysing phosphoric acid monoesters into a phosphate ion and a molecule with a free hydroxyl group (see dephosphorylation).
Fosfluconazole was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on Oct 16, 2003. It was developed and marketed as Prodif® by Pfizer in Japan.
Fosfluconazole is a water-soluble phosphate prodrug of fluconazole – a triazole antifungal drug. It is indicated for the treatment of candida and cryptococcus infections.
Prodif® is available as solution for intravenous use, containing 100, 200 or 400 mg of free Fosfluconazole per vial. The recommended dose is 50 to 100 mg administered intravenously once daily for candidiasis. Another dose is 50 to 200 mg fluconazole once daily for cryptococcosis.


Reference:1. WO9728169A1 / US6977302B2.
2. Org. Process Res. Dev.2002, 6, 109-112.
http://pubs.acs.org/doi/pdf/10.1021/op010064%2B
2-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazole-1-yl)- 2-propyl dihydrogen phosphate (2). A slurry of dibenzyl 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazole-1-yl)-2-propyl phosphate (30.1 kg, 53.13 mol), 5% palladium-on-carbon catalyst (50% wet, type 5R39, 1.5 kg), and sodium hydroxide (4.36 kg, 108.9 mol) in low-endotoxin water (75.7 L) was hydrogenated at ambient temperature and 414 kPa (60 psi) for 12 h. The slurry was filtered, and the catalyst was washed with low-endotoxin water (9.8 L). After separating the toluene by-product, the aqueous phase was slurried with carbon (3.1 kg) for 30 min. After the carbon was removed by filtration, the aqueous phase was acidified to pH 1.45 by that addition of sulfuric acid (6.69 kg) in low-endotoxin water (25 L) over 2 h. The resulting slurry was granulated at ambient temperature for 1 h and then filtered. The product was sequentially washed with filtered low-endotoxin water (103 L) and filtered acetone (103 L). The product was dried under vacuum at 50 °C for 12 h to give the title compound (18.1 kg, 88%) a white powder: mp 223-224 °C.
1H NMR (DMSO) δ 5.07 (2H, d), 5.24 (2H, d), 6.77-6.83 (1H, m), 7.00-7.18 (2H, m), 7.75 (2H, s), 8.53 (2H, s).
Found: C, 40.28; H, 3.39; N, 21.63;
[MH]+ 387.0786. C13H13F2N6O4P requires: C, 40.43; H, 3.39; N, 21.78; [MH]+ 387.0782.
US6977302
https://www.google.com/patents/US6977302
EXAMPLE 1 1-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propyl dihydrogen phosphate
(a) Dibenzyl 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propyl phosphate
Method A
A solution of 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)propan-2-ol (also known as fluconazole, 10.0 g, 32.6 mmol), 1H-tetrazole (6.85 g, 97.8 mmol), dibenzyl diisopropyl phosphoramidite (22.55 g, 65.2 mmol) in methylene chloride (100 ml) was stirred at room temperature under a nitrogen atmosphere for 2 hours. The mixture was then cooled to 0° C., and a solution of 3-chloroperoxybenzoic acid (13.5 g, 50-55% w/w, 39.1 mmol) in methylene chloride (50 ml) was added maintaining the temperature at 0° C. The resulting mixture was allowed to warm to room temperature for 1 hour before washing with aqueous sodium metabisulphite and sodium bicarbonate. After drying (MgSO4) the solvent was removed and replaced with methyl isobutyl ketone (37 ml) and tert-butyl methyl ether (74 ml). After granulating at −10° C. for 1 hour the product was filtered and washed with ice cold methyl isobutyl ketone and tert-butyl methyl ether (1:3, 15 ml) and dried at 50° C. under vacuum for 18 hours to give the subtitle compound (16.05 g, 87%), m.p. 93° C.
Found: C, 57.12; H, 4.46; N, 14.85. C27H25F2N6O4P requires C, 57.24; H, 4.46; N, 14.84%. m/z 567 (MH+) 1H NMR (300 MHz, CDCl3) δ=4.90 (d, 2H), 4.95 (d, 2H), 5.05 (d, 2H), 5.19 (d, 2H), 6.58-6.73 (m, 2H), 6.88-6.95 (m, 1H), 7.20-7.30 (m, 4H) 7.32-7.38 (m; 6H), 7.80 (s, 2H), 8.36 (s, 2H).
Method B
To stirred ethyl acetate (1530 ml) was added 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)propan-2-ol (also known as fluconazole, 306 g, 1.00 mol) and pyridine (237.3 g, 3.00 mol) before cooling to 0° C. Phosphorus trichloride (137.4 g, 1.00 mol) was added dropwise to the reaction mixture maintaining the temperature between 0-5° C. before allowing the reaction mixture to warm to 15° C. over 30 minutes. Benzyl alcohol (216 g, 2.00 mol) was then added over 30 minutes at 15-20° C. After a further 30 minutes hydrogen peroxide (27.5% w/w in water, 373 g) was added maintaining the temperature at 15-20° C. After 30 minutes the aqueous phase was removed and the organic phase washed with aqueous sodium metabisulphite, dilute hydrochloric acid and water. The solvent was removed at reduced pressure and replaced with methyl isobutyl ketone (850 ml) and tert-butyl methyl ether (1132 ml). After granulating at 20° C. for 1 hour and at 0° C. for 1 hour, the product was filtered and washed with ice cold tert-butyl methyl ether (2×220 ml) and dried at 50° C. under vacuum for 18 hours to give the subtitle compound (358 g, 63%). The melting point and spectroscopic data was identical to that stated in method A.
(b) 2-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propyl dihydrogen phosphate
A slurry of the compound of step (a) (9.80 g, 17.3 mmol), 5% palladium on carbon catalyst (50% wet, 1.0 g) and sodium hydroxide (1.38 g, 34.6 mmol) in water (26 ml) was hydrogenated at room temperature and 414 kPa (60 p.s.i.) for 20 hours. The solution was filtered through a pad of celite (trade mark) and washed with water (5 ml). The toluene was separated and the aqueous phase cooled to 0° C. whereupon sulphuric acid (1.70 g, 17.3 mmol) was added. The resulting slurry was granulated at 0° C. for 1 hour and then filtered, washed with water (2×5 ml) and dried under vacuum at 50° C. to give the title compound (5.80 g, 87%). m.p. 223-224° C.
Found: C, 40.28; H, 3.39; N, 21.63. C13H13F2N6O4P requires C, 40.43; H, 3.39; N, 21.76%. 1H NMR (300 MHz, DMSO) δ=5.07 (d, 2H) 5.24 (d, 2H), 6.77-6.83 (m, 1H), 7.00-7.18 (m, 2H), 7.75 (s, 2H), 8.53 (s, 2H).
EXAMPLE 2 2-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propyl disodium phosphate
A solution of the compound of Example 1(a) (10.0 g, 17.7 mmol) and sodium acetate (2.90 g, 35.3 mmol) in ethanol (160 ml) and water (20 ml) was hydrogenated over Pearlman’s catalyst (1.00 g) at room temperature and at 345 kPa (50 p.s.i.) for 16 hours. The solution was filtered through a pad of celite (trade mark) and the solvents removed at reduced pressure to leave a thick syrup. This was dissolved in ethanol (100 ml) with the aid of sonication and warmed to reflux. The resulting solution was allowed to cool slowly and granulate for 1 hour at room temperature. The product was filtered, washed with ethanol (10 ml) and dried under vacuum at 50° C. to give the title compound (4.48 g, 59%). m.p. 160-162° C.
1H NMR (300 MHz, D2O) δ=5.01 (d, 2H), 5.40 (d, 2H), 6.60 (m, 1H), 6.79 (m, 1H), 7.11 (m, 1H), 7.63 (s, 2H), 8.68 (s, 2H).
Route 2
![]()
Reference:1. CN103864844A.
http://www.google.com/patents/CN103864844A?cl=en
TRANSLATED BY MACHINE…….TEXT MAY VARY
forskolin fluconazole (fosf Iuconazole, Formula I) is fluconazole (Formula IV) of monophosphate prodrugs, fluconazole in the tertiary alcohol into a phosphate ester, not only did not introduce a chiral center, also increased water solubility, because a long time to overcome the low water solubility of fluconazole resulting larger infusion volume defects. After intravenous administration in the role of phosphatases in vivo hydrolysis into fluconazole, pharmacological effect. Blessing from the Central Institute of the United States Secretary of fluconazole Fai end developed, launched in Japan in 2004 I May 15, for the treatment of candidiasis and cryptococcal infections caused deep as true bacteremia, respiratory fungal disease, fungal peritoneum
Inflammation, gastrointestinal fungal disease, fungal urinary tract infections, fungal meningitis.
Synthesis gas itraconazole on forskolin in W09728169, Organic Process Research & Development (200 2), 6 (2), 109-112, CN1789270, Art of Drug Synthesis (2007), 71-82, etc. have been reported in the literature . Which Organic Process Research & Development (2002) described in detail in the first blessing Secretary fluconazole and improved synthetic route for the route problems to adapt to industrial mass production of synthetic routes.
Document Organic Process Research & Development (2002), 6,109-112 discloses the following two synthetic routes.
Route One:
Route two:
The final step is a route to the removal of benzyl group in a methanol solvent by palladium on carbon catalyzed hydrogenation reaction yield was 65%. Since forskolin fluconazole final product insoluble in methanol, and therefore there is a route following shortcomings: a catalyst poisoning, the final product is easy to form methanol solvate, removing the catalyst in the loss of product, the final product are difficult to separate, low yield not suitable for industrial production.
Two routes still using palladium on carbon hydrogenation debenzylation, except that the solvent was changed to sodium hydroxide solution, the product of soluble and stable in aqueous sodium hydroxide solution, after filtering off the catalyst, forskolin fluoro itraconazole by acidification of sodium sulfate can be easily obtained blessing Secretary of fluconazole, the reaction yield of 85-90%.
In the prior art, the removal of benzyl preparation blessing Secretary of fluconazole, the use of a pressure hydrogenation, relatively harsh reaction conditions; and blessing Secretary of fluconazole in water and slightly soluble in methanol, for blessing Secretary fluconazole further refined and purified more difficult. The present invention aims to provide a new and suitable for industrial production methods blessing Secretary fluconazole.
Example 1
2- (2,4-gas-phenyl) -1,3-bis (1H-1, 2,4- two P sat 1-yl) -2-propyl-di-benzyl-pity Cool ( Preparation blessing Secretary fluconazole dibenzyl ester)
Step The method according to CN1210540A in Example 1 A or Method B of (a), was prepared to give the title compound, having 1H-NMR shown in Figure 1 (SOi) MHz, DMS0-D6) spectrum.
Example 2
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas
Itraconazole ammonium salt) Preparation
Formula III blessing Secretary fluconazole two benzyl ester (566g, lmol), 120g of dry Pd / C (containing 5% palladium) and ammonium formate (315g, 5mol) in methanol (6L), and stirred under reflux for 5h , TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (566ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 415g, yield 98.8%.
] lH-Mffi (500MHz, DMS0-D6) δ: 4.87-4.90, 5.58-5.61,6.56-6.60, 6.94-7.03,7.52-7.61,8.96, having 1H-NMR shown in Figure 2 (500MHz, DMS0 -D6) spectrum.
Example 3
2- (2,4-gas-phenyl) -1,3-bis (1H-1, 2,4- two 1-yl) -2-propyl-pity acid dioxide Cool (forskolin
Fluconazole) Preparation of
[0052] Formula II forskolin fluconazole salt (420g, Imol), in water (IL) while stirring, filtered, 2mol / L sulfuric acid aqueous solution (500ml), 5 ° C under stirring for lh, filtered, cold water ( 200ml) wash, 50 ° C under dry blessed Division fluconazole 379g, yield 98%.
1H-Mffi (SOOMHz) DMSO-De) δ:. 5.09-5.12,5.25-5.28,6.80-6.84,7.05-7.16,7.77,8.55,10.32 [0054] Example 4
2_ (2,4_ two gas-phenyl) -1, double 3_ (1Η-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (566g, lmol), 84g of dry Pd / C (5% containing button) and ammonium formate (189g, 3mol) in anhydrous methanol (5L) in the mixture was stirred at reflux for 5h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 410g, yield 97.5%.
Example 5
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (566g, lmol), 30g of dry Pd / C (containing 10% palladium) and ammonium formate (315g, 5mol) in anhydrous methanol (5L) in the mixture was stirred at reflux for 5h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 405g, yield 96.4%.
Example 6
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (566g, lmol), 30g of dry Pd / C (containing 10% palladium) and ammonium formate (315g, 5mol) in ethanol (12L) and stirred was refluxed for 5h, TLC monitoring completion of the reaction, was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 395g, 94% yield.
Example 7
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
forskolin fluconazole dibenzyl ester (566g, lmol), 170g of dry Pd / C (containing 5% of palladium) and ammonium formate (315g, 5mol) in ethanol (16L) was stirred under reflux for 5h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 398g, yield 94.7%.
Example 8
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (566g, lmol), 120g of dry Pd / C (containing 5% palladium) and ammonium formate (315g, 5mol) in isopropanol (12L) in the mixture was stirred at reflux for 5h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 402g, a yield of 95.7%.
Example 9
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
[0071] under nitrogen blessing Secretary fluconazole dibenzyl ester (566g, lmol), 60g of dry Pd / C (containing 5% palladium) and ammonium formate (504g, 8mol) in methanol (8L) in, 50 ° C under stirring reaction 40h, TLC monitoring completion of the reaction, was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added ^ OOml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 398g, yield 94.8%.
Example 10
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (5668,111101), 8 (^ dry? (1 / (:( containing palladium 5%) and ammonium formate (315g, 5mol) for n-propyl alcohol (12L) in, 60 ° C the reaction was stirred 20h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 398g 95% yield.
Example 11
2- (2,4-gas-phenyl) -1,3-bis (1H-1, 2,4- sit two P-1-yl) -2-propyl-pity acid dioxide Cool (forskolin fluconazole) Preparation of [0077] under nitrogen blessing Secretary fluconazole dibenzyl ester 566 g (Imol) adding 56g of dry Pd / C (containing 5% palladium), methanol 6L, 315 g of ammonium formate, stirring boil under reflux for 5h, TLC after completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, addition of IL of water and dissolved with stirring, filtered, 2mol / L sulfuric acid 500mL, 5 ° C with stirring to precipitate lh, filtered, 200mL cold water, 50 ° C drying 365 g, 95% yield.
Example 12 forskolin fluconazole salt and HPLC detection methods blessing Secretary fluconazole:
High performance liquid chromatography (Chinese Pharmacopoeia 2010 edition two Appendix VD): octadecylsilane bonded silica as a filler, Column: Thermo BDS C18 (4.6 X 150mm, 3.5 μ m); methanol as mobile phase A, phosphate buffer (take potassium dihydrogen phosphate 0.68g, set 1000ml water, triethylamine 6ml, adjusted to pH 5.0 with phosphoric acid) as the mobile phase B, a flow rate of 1.0ml / min; column temperature 35 ° C; detection wavelength was 210nm, linear gradient.
After the examination, according to the peak area calculation, purity prepared in Example 2-11 was the implementation of the target product of 99.5%.
| Patent | Submitted | Granted |
|---|---|---|
| Nanoparticulate Anidulafungin Compositions and Methods for Making the Same [US2009238867] | 2009-09-24 | |
| IMIDAZOPYRIDINE SUBSTITUTED TROPANE DERIVATIVES WITH CCR5 RECEPTOR ANTAGONIST ACTIVITY FOR THE TREATMENT OF HIV AND INFLAMMATION [US7790740] | 2008-02-21 | 2010-09-07 |
| Pharmaceutical formulations of cyclodextrins and antifungal azole compounds [US2007082870] | 2007-04-12 | |
| TRIAZOLE DERIVATIVES USEFUL IN THERAPY [EP0880533] | 1998-12-02 | 2002-06-12 |
| Triazole derivatives useful in therapy [US6790957] | 2003-07-31 | 2004-09-14 |
| Process for controlling the hydrate mix of a compound [US7323572] | 2004-01-15 | 2008-01-29 |
| TOPICAL TERBINAFINE FORMULATIONS AND METHODS OF ADMINISTERING SAME FOR THE TREATMENT OF FUNGAL INFECTIONS [US7820720] | 2010-04-29 | 2010-10-26 |
| PHARMACEUTICAL COMPOSITION COMPRISING PHENYLAMIDINE DERIVATIVE AND METHOD OF USING THE PHARMACEUTICAL COMPOSITION IN COMBINATION WITH ANTIFUNGAL AGENT [US8173157] | 2010-04-22 | 2012-05-08 |
| COMPOSITIONS COMPRISING POLYUNSATURATED FATTY ACID MONOGLYCERIDES OR DERIVATIVES THEREOF AND USES THEREOF [US8222295] | 2009-11-26 | 2012-07-17 |
| MASKED CARBOXYLATE NEOPENTYL SULFONYL ESTER CYCLIZATION RELEASE PRODRUGS OF ACAMPROSATE, COMPOSITIONS THEREOF, AND METHODS OF USE [US2009069419] | 2009-03-12 |
| Patent | Submitted | Granted |
|---|---|---|
| Triazole derivatives useful in therapy [US2005130940] | 2005-06-16 | |
| Chemical compounds [US7309790] | 2005-06-16 | 2007-12-18 |
| Combination of voriconazole and an antifungal CYP2C19 inhibitor [US2005182074] | 2005-08-18 | |
| Inhibitors of fungal invasion [US2004106663] | 2004-06-03 | |
| Triazole derivatives useful in therapy [US6977302] | 2004-11-25 | 2005-12-20 |
| Pharmaceuticals [US7691877] | 2007-08-23 | 2010-04-06 |
| SIMPLE PANTOIC ACID ESTER NEOPENTYL SULFONYL ESTER CYCLIZATION RELEASE PRODRUGS OF ACAMPROSATE, COMPOSITIONS THEREOF, AND METHODS OF USE [US7994218] | 2009-03-26 | 2011-08-09 |
| COMPLEX PANTOIC ACID ESTER NEOPENTYL SULFONYL ESTER CYCLIZATION RELEASE PRODRUGS OF ACAMPROSATE, COMPOSITIONS THEREOF, AND METHODS OF USE [US8168617] | 2009-03-19 | 2012-05-01 |
| Purine derivatives [US7642350] | 2006-11-23 | 2010-01-05 |
| IMIDAZOPYRIDINONES [US2009221631] | 2009-09-03 |
IMPURITIES
1
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
{[2-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)propan-2-yl]oxy}phosphonic acid
|
|
| Clinical data | |
| AHFS/Drugs.com | International Drug Names |
| Legal status |
|
| Routes of administration |
IV |
| Identifiers | |
| CAS Number | 194798-83-9  |
| ATC code | None |
| PubChem | CID 214356 |
| ChemSpider | 185843  |
| UNII | 3JIJ299EWH  |
| ChEMBL | CHEMBL1908301  |
| Chemical data | |
| Formula | C13H13F2N6O4P |
| Molar mass | 386.25 g/mol |
| CN1210540A * | Jan 27, 1997 | Mar 10, 1999 | 辉瑞研究开发公司 | Triazole derivatives useful in therapy |
| CN1789270A * | Dec 16, 2005 | Jun 21, 2006 | 西安新安医药科技有限公司 | Mycotic ingection-resisting fosfluconazole hydrate and preparation method thereof |
| CN101890028A * | Feb 22, 2007 | Nov 24, 2010 | 卫材R&D管理有限公司 | Stabilized pharmaceutical composition |
| CN102439018A * | Mar 3, 2010 | May 2, 2012 | 塞普斯制药有限公司 | Fosfluconazole derivatives, synthesis, and use in long acting formulations |
| US20040007689 * | Jun 23, 2003 | Jan 15, 2004 | Pfizer Inc. | Process for controlling the hydrate mix of a compound |
| Reference | ||||
|---|---|---|---|---|
| 1 | * | ARTHUR BENTLEY等: “The Discovery and Process Development of a Commercial Route to the Water Soluble Prodrug, Fosfluconazole“, 《ORGANIC PROCESS RESEARCH & DEVELOPMENT》, vol. 6, no. 2, 18 December 2001 (2001-12-18), XP002491526, DOI: doi:10.1021/op010064+ | ||
| 2 | * | 国大亮 等: “福司氟康唑“, 《齐鲁药事》, vol. 24, no. 1, 30 January 2005 (2005-01-30), pages 60 | ||
| 3 | * | 村上尚道: “fosfluconazole“, 《NEW DRUGS OF THE WORLD:2003》, vol. 33, no. 10, 15 September 2004 (2004-09-15), pages 56 | ||
//////UK-292663, UK 292663, F-FLCZ, F FLCZ, Fosfluconazole, 194798-83-9, UNII-3JIJ299EWH, 3JIJ299EWH, NCGC00182029-01
Fc1ccc(c(F)c1)C(OP(=O)(O)O)(Cn2ncnc2)Cn3ncnc3
Filed under: Japan marketing, Japan pipeline Tagged: 194798-83-9, 3JIJ299EWH, F-FLCZ, Fosfluconazole, JAPAN, NCGC00182029-01, PFIZER, UK-292663, UNII-3JIJ299EWH