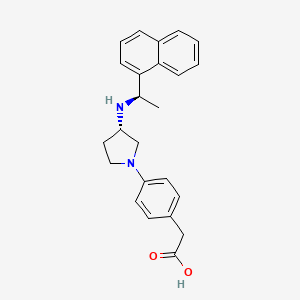
Evocalcet
C24H26N2O2, 374.484
Evocalcet; UNII-E58MLH082P; E58MLH082P; 870964-67-3; Evocalcet [INN]; Orkedia (TN)
エボカルセト
Эвокальцет [Russian] [INN]
エボカルセト
Evocalcet

C24H26N2O2 : 374.48
[870964-67-3]
KHK 7580 …..example
| 3.008 | 2HCl | MS · APCI: 375[M + H]+ |
in EP1757582
4-(3S-(1R-(1-naphthyl)ethylamino)pyrrolidin-1- yl)phenylacetic acid
4-[(3S)-3-[[(1R)-1-(1-naphthalenyl)ethyl]amino]-1-pyrrolidinyl]-Benzeneacetic acid,
BASE ….870964-67-3
DI HCL SALT …….870856-31-8
MF C24 H26 N2 O2 BASE
MW 374.48 BASE
KHK-7580
KHK-7580; MT-4580
Mitsubishi Tanabe Pharma Corp… innovator
Kyowa Hakko Kirin Co Ltd.. licencee
4-(3S-(1R-(1-naphthyl)ethylamino)pyrrolidin-1-yl)phenylacetic acid,
Evocalcet (trade name Orkedia) is a drug for the treatment of hyperparathyroidism.[1] It acts as a calcium-sensing receptor agonist.[2]
In 2018, it was approved in Japan for treatment of secondary hyperparathyroidism in patients on dialysis.[3]
useful as calcium-sensitive receptor (CaSR) agonists for treating hyperparathyroidism. a CaSR agonist, being developed by Kyowa Hakko Kirin, under license from Mitsubishi Tanabe, for treating secondary hyperparathyroidism (phase 2 clinical, as of March 2015).
WO2005115975,/EP1757582
http://www.google.co.in/patents/EP1757582A1?cl=en
Example no
| 3.008 | 2HCl | MS · APCI: 375[M + H]+ |
Mitsubishi Tanabe Pharma Corporation
The present invention provides a novel crystal form of an arylalkylamine
compound. Specifically, a novel crystal form of
4-(3S-(1R-(1-naphthyl)ethylamino)pyrrolidin-1- yl)phenylacetic acid has
excellent stability, and is therefore useful as an active ingredient for
a medicine. The present invention also provides an industrially
advantageous method for producing an arylalkylamine compound.
http://worldwide.espacenet.com/publicationDetails/biblio?DB=worldwide.espacenet.com&II=0&ND=3&adjacent=true&locale=en_EP&FT=D&date=20150312&CC=WO&NR=2015034031A1&KC=A1
Mitsubishi Tanabe Pharma Corporation
The present invention provides a novel crystal form of an arylalkylamine compound. Specifically, a novel crystal form of 4-(3S-(1R-(1-naphthyl)ethylamino)pyrrolidin-1- yl)phenylacetic acid has excellent stability, and is therefore useful as an active ingredient for a medicine. The present invention also provides an industrially advantageous method for producing an arylalkylamine compound.
PATENT
Reference Example 3.001
(1) To a mixed solution containing 33.5 g of 3-hydroxypiperidine and 62.7 ml of triethylamine dissolved in 250 ml of methylene chloride was added dropwise a solution of 55.7 ml of benzyloxycarbonyl chloride in 150 ml of methylene chloride, and the mixture was stirred at room temperature for 16 hours. To the reaction mixture were added a saturated aqueous citric acid and chloroform, the mixture was stirred and the liquids were separated. The organic layer was dried, the solvent was evaporated, and the residue was purified by silica gel column chromatography (hexane:ethyl acetate=4:1→0:1) to obtain 75.5 g of benzyl 3-hydroxypiperidine-1-carboxylate.
MS•APCI (m/z): 236 [M+H]+
(2) 800 ml of a solution of 52.4 ml of oxalyl chloride in methylene chloride was cooled to −78° C., 53.2 ml of DMSO was added dropwise to the solution, and the mixture was stirred at −78° C. for 0.5 hour. A solution of 75.5 g of benzyl 3-hydroxypiperidine-1-carboxylate dissolved in 200 ml of methylene chloride was added dropwise to the mixture, and further 293 ml of triethylamine was added dropwise to the same, and the mixture was stirred for 16 hours while a temperature thereof was gradually raised to room temperature. To the reaction mixture were added a saturated aqueous sodium bicarbonate solution and chloroform, the mixture was stirred and the liquids were separated. The organic layer was dried and concentrated to obtain 83.7 g of 1-benzyloxycarbonyl-3-piperidone. MS•APCI (m/z): 234 [M+H]+
(3) To a solution of 83.7 g of 1-benzyloxycarbonyl-3-piperidone dissolved in 1.2 liters of methylene chloride was added 55.0 g of (R)-(+)-1-(1-naphthyl)ethylamine, and after the mixture was stirred at room temperature for 2 hours, 69 ml of acetic acid and 160 g of sodium triacetoxy borohydride were added to the mixture, and the mixture was stirred at room temperature for 15 hours. To the reaction mixture was added an aqueous sodium hydroxide to make the mixture basic, and then, chloroform was added to the mixture, the mixture was stirred and the liquids were separated. The organic layer was dried and concentrated, and the residue was purified by silica gel column chromatography (hexane:ethyl acetate=4:1→0:1) to obtain 98.7 g of benzyl 3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate. MS•APCI (m/z): 389 [M+H]+
(4) To a solution of 40.95 g of triphosgene dissolved in 800 ml of methylene chloride was added dropwise a mixed solution containing 80.6 g of benzyl 3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate and 86.6 ml of triethylamine dissolved in 200 ml of methylene chloride at 0° C., and the mixture was stirred at room temperature for 16 hours. To the reaction mixture was added water, the mixture was stirred and the liquids were separated. The organic layer was dried and concentrated, and the residue was washed with 200 ml of diethyl ether, and the crystal collected by filtration was recrystallized from chloroform and diethyl ether to obtain 48.9 g of benzyl (R)-3-[chlorocarbonyl-(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate.
Further, the filtrate was purified by silica gel column chromatography (hexane:ethyl acetate=8:1→0:1) to obtain 5.82 g of benzyl (R)-3-[chlorocarbonyl-(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate and 14.5 g of benzyl (S)-3-[chlorocarbonyl-(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate.
(5) To a solution containing 54.6 g of benzyl (R)-3-[chlorocarbonyl-(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate dissolved in 700 ml of tetrahydrofuran was added 350 ml of water, and the mixture was stirred under reflux for 15 hours. After tetrahydrofuran was evaporated, a saturated aqueous sodium bicarbonate solution and chloroform were added thereto, the mixture was stirred and the liquids were separated. The organic layer was dried and concentrated, and the residue was purified by silica gel column chromatography (hexane:ethyl acetate=4:1→0:1) to obtain 24.3 g of benzyl (R)-3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate. MS•APCI (m/z): 389 [M+H]+
(6) To a solution containing 24.2 g of benzyl (R)-3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate dissolved in 250 ml of methanol was added 2.5 g of palladium carbon (10% wet), and the mixture was shaked under hydrogen atmosphere at 3 atm at room temperature for 40 hours. Palladium carbon was removed, and the solvent was evaporated, the residue was washed with ethyl acetate-chloroform (10:1), and collected by filtration to obtain 15.3 g of (R)-3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine (the following Reference example Table, Reference example 3.001(a)). MS•APCI (m/z): 255 [M+H]+
(7) By using 14.5 g of benzyl (S)-3-[chlorocarbonyl-(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate, the same treatment was carried out as in the above-mentioned (5) to obtain 4.74 g of benzyl (S)-3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate. MS•APCI (m/z): 389 [M+H]+
Moreover, by using 4.7 g of benzyl (S)-3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine-1-carboxylate, the same treatment was carried out as in the above-mentioned (6) to obtain 2.89 g of (S)-3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine. MS•APCI (m/z): 255 [M+H]+
(8) To a solution of 3.46 g of (S)-3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine dissolved in 15 ml of methanol was added dropwise 20 ml of a solution of 4M hydrochloric acid in ethyl acetate, and the mixture was stirred. The reaction mixture was concentrated under reduced pressure, diethyl ether was added to the residue, washed and dried to obtain 3.33 g of (S)-3-[(R)-1-(naphthalen-1-yl)ethylamino]piperidine dihydrochloride
| 3.008 | 2HCl | MS · APCI: 375[M + H]+ |
| TABLE A3 | |||||
| Example No. | R1—X— | —Ar | Salt | Physical properties, etc. | |
CLIP
see all at http://drugpatentsint.blogspot.in/2015/03/wo-2015034031.html
see all at http://drugpatentsint.blogspot.in/2015/03/wo-2015034031.html
see all at http://drugpatentsint.blogspot.in/2015/03/wo-2015034031.html
see all at http://drugpatentsint.blogspot.in/2015/03/wo-2015034031.html
see all at http://drugpatentsint.blogspot.in/2015/03/wo-2015034031.html
do not miss out on above click
http://www.kyowa-kirin.com/research_and_development/pipeline/
KHK7580 -Secondary Hyperparathyroidism




JP
| Company | Mitsubishi Tanabe Pharma Corp. |
| Description | Calcium receptor agonist |
| Molecular Target | |
| Mechanism of Action | Calcium-sensing receptor (CaSR) agonist |
| Therapeutic Modality | Small molecule |
| Latest Stage of Development | Phase II |
| Standard Indication | Thyroid disease |
| Indication Details | Treat hyperparathyroidism in patients receiving hemodialysis; Treat secondary hyperparathyroidism (SHPT) |
| Regulatory Designation | |
| Partner |
August 29, 2014
Kyowa Hakko Kirin Announces Commencement of Phase 2b Clinical Study of KHK7580 in Patients with Secondary Hyperparathyroidism in Japan
Tokyo, Japan, August 29, 2014 — Kyowa Hakko Kirin Co., Ltd. (Tokyo: 4151, President and CEO: Nobuo Hanai, “Kyowa Hakko Kirin”) today announced the initiation of a phase 2b clinical study evaluating KHK7580 for secondary hyperparathyroidism patients receiving hemodialysis in Japan.
This randomized, placebo-controlled, double-blind, parallel-group, multi-center study is designed to evaluate efficacy and safety in cohorts comprising KHK7580, its placebo and cinacalcet and initial dose of KHK7580 for secondary hyperparathyroidism patients receiving hemodialysis.
KHK7580 is a small molecular compound produced by Mitsubishi Tanabe Pharma Corporation (President & Representative Director, CEO: Masayuki Mitsuka, “Mitsubishi Tanabe Pharma”). Kyowa Hakko Kirin signed a license agreement of KHK7580 with Mitsubishi Tanabe Pharma for the rights to cooperative research, develop, market and manufacture the product in Japan and some part of Asia on March 2008.
The Kyowa Hakko Kirin Group is contributing to the health and prosperity of the world’s people by pursuing advances in life sciences and technology and creating new value.
Outline of this study
| CLINICALTRIALS.GOV IDENTIFIER |  NCT02216656 NCT02216656 |
|---|---|
| TARGET POPULATION | Secondary hyperparathyroidism patients receiving hemodialysis |
| TRIAL DESIGN | Randomized, placebo-controlled, double-blind (included open arm of cinacalcet), parallel-group, multi-center study |
| ADMINISTRATION GROUP | KHK7580, Placebo, cinacalcet |
| TARGET NUMBER OF SUBJECTS | 150 |
| PRIMARY OBJECTIVE | Efficacy |
| TRIAL LOCATION | Japan |
| TRIAL DURATION | Jul. 2014 to Jun. 2015 |
Contact:
Kyowa Hakko Kirin
Media Contact:
+81-3-3282-1903
or
Investors:
+81-3-3282-0009
Update on march 2016
New comment waiting approval on New Drug Approvals |
|
http://www.medkoo.com/products/6729

Name: Evocalcet
CAS#: 870964-67-3
Chemical Formula: C24H26N2O2
Exact Mass: 374.19943
Evocalcet is a calcium-sensing receptor agonist. The calcium-sensing receptor (CaSR) is a Class C G-protein coupled receptor which senses extracellular levels of calcium ion. The calcium-sensing receptor controls calcium homeostasis by regulating the release of parathyroid hormone (PTH). CaSR is expressed in all of the organs of the digestive system. CaSR plays a key role in gastrointestinal physiological function and in the occurrence of digestive disease. High dietary Ca2+ may stimulate CaSR activation and could both inhibit tumor development and increase the chemotherapeutic sensitivity of cancer cells in colon cancer tissues. (Last update: 12/15/2015).
Synonym: MT-4580; MT 4580; MT4580; KHK-7580; KHK7580; KHK 7580; Evocalcet
IUPAC/Chemical Name: 2-(4-((S)-3-(((R)-1-(naphthalen-1-yl)ethyl)amino)pyrrolidin-1-yl)phenyl)acetic acid
2
https://tripod.nih.gov/ginas/app/substance/f580b9fd
http://www.drugspider.com/drug/evocalcet
| INN NAME |
Evocalcet
|
||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LAB CODE(S) |
MT-4580
KHK-7580
|
||||||||||||||||||||||||||||||||||||||||
| CHEMICAL NAME |
{4-[(3S)-3-{[(1R)-1-(Naphthalen-1-yl)ethyl]amino}pyrrolidin-1-yl]phenyl}acetic acid
|
||||||||||||||||||||||||||||||||||||||||
| CHEMICAL STRUCTURE |
 |
||||||||||||||||||||||||||||||||||||||||
| MOLECULAR FORMULA |
C24H26N2O2
|
||||||||||||||||||||||||||||||||||||||||
| SMILES |
O=C(O)CC1=CC=C(N2C[C@@H](N[C@@H](C3=C4C=CC=CC4=CC=C3)C)CC2)C=C1
|
||||||||||||||||||||||||||||||||||||||||
| CAS REGISTRY NUMBER |
870964-67-3
|
||||||||||||||||||||||||||||||||||||||||
| ORPHAN DRUG STATUS |
No
|
||||||||||||||||||||||||||||||||||||||||
| ON FAST TRACK |
No
|
||||||||||||||||||||||||||||||||||||||||
| NEW MOLECULAR ENTITY |
Yes
|
||||||||||||||||||||||||||||||||||||||||
| ORIGINATOR | |||||||||||||||||||||||||||||||||||||||||
| DEVELOPER(S) | |||||||||||||||||||||||||||||||||||||||||
| CLASS | |||||||||||||||||||||||||||||||||||||||||
| MECHANISM OF ACTION | |||||||||||||||||||||||||||||||||||||||||
| WHO ATC CODE(S) | |||||||||||||||||||||||||||||||||||||||||
| EPHMRA CODE(S) | |||||||||||||||||||||||||||||||||||||||||
| CLINICAL TRIAL(S) |
|
||||||||||||||||||||||||||||||||||||||||
| UPDATED ON |
11 Oct 2015
|
CLIP
https://www.sciencedirect.com/science/article/pii/S0960894X18303676


Scheme 1. Synthesis of key intermediates S4, S5, S9, and S10. Reagents and conditions: (a) Tf2O, i-Pr2NEt, CH2Cl2, −20 °C. Then, (R)-(+)-1-(1-naphthyl)ethylamine, −20 °C to rt (S1 57%); (b) triphosgene, Et3N, CH2Cl2, −20 °C to 5 °C. Then, i-Pr2NEt, tert-butanol, 70 °C; (c) separation via silica gel chromatography (S2 31%, S3 33% in 2 steps); (d) HCl, chloroform, 1,4-dioxane, rt (S4 > 94%, S5 > 94%); (e) (R)-(+)-1-(1-naphthyl)ethylamine, NaBH(OAc)3, acetic acid, CH2Cl2, rt (S6 79%); (f) triphosgene, Et3N, CH2Cl2, 0 °C to rt; (g) separation via filtration and silica gel chromatography (S7 58%, S8 16% in 2 steps); (h) water, tetrahydrofuran, reflux; (i) H2, Pd/C, methanol, rt (S9 50% in 2 steps); (j) HCl, ethyl acetate, methanol, rt (S10 31% in 3steps).

Scheme 2. Synthesis of 2–15 and evocalcet (16). Reagents and conditions: (a) aryl iodide or aryl bromide, palladium acetate, (rac)-BINAP, sodium tert-butoxide, toluene, 80 °C or reflux; (b) HCl, ethyl acetate or 1,4-dioxane, rt; (c) tert-butyl 4-fluorobenzoate, potassium carbonate, DMSO, 130 °C; (d) HCl, 1,4-dioxane, 45 °C; (e) 2-aminoethanol, EDC hydrochloride, HOBt, Et3N, DMF, rt; (f) 5-(4-bromophenyl)-2-(triphenylmethyl)–2H-tetrazole, Pd2(dba)3, (2-biphenyl)di-tert-butylphosphine, sodium tert-butoxide, toluene, rt; (g) HCl, water, 1,4-dioxane, rt; (h) tert-butyl 4-bromobenzoate, palladium acetate, (rac)-BINAP, sodium tert-butoxide, toluene, reflux; (i) trifluoroacetic acid, rt. Then, HCl, ethyl acetate or 1,4-dioxane, rt (2 23%, 3 21%, 4 44%, 5 34%, 620%, 7 55%, 8 29%, 9 21%, 10 19%, 11 40%, 13 26%, 14 69%, 15 69% in 2 steps); (j) 3-(trifluoromethoxy)phenylboronic acid, copper acetate, Et3N, CH2Cl2, molecular sieve 4A, rt (S117%); (k) (COCl)2, DMSO, Et3N, CH2Cl2, −60 °C to rt (S12 was used in the next step without purification); (l) (R)-(+)-1-(1-naphthyl)ethylamine, NaBH(OAc)3, acetic acid, CH2Cl2, rt. Then, separation of isomers; (m) HCl, ethyl acetate, rt (12 10% in 2 steps); (n) ethyl 4-bromophenylacetate, Pd2(dba)3, (2-biphenyl)di-tert-butylphosphine, sodium tert-butoxide, toluene, rt (S13 63%); (o) aqueous sodium hydroxide solution, ethanol, rt (evocalcet (16) 73%).

evocalcet as a white crystal. MS-APCI (m/z): 375 [M+H]+ .
1H NMR (400 MHz, DMSO-d6) δ 8.25-–8.37 (m, 1H), 7.88–7.97 (m, 1H), 7.79 (d, J = 7.9 Hz, 1H), 7.74 (d, J = 6.9 Hz, 1H), 7.39–7.57 (m, 3H), 7.01 (d, J = 8.6 Hz, 2H), 6.38 (d, J = 8.6 Hz, 2H), 4.74 (q, J = 6.4 Hz, 1H), 3.37 (s, 2H), 3.18–3.34 (m, 3H), 3.03–3.15 (m, 1H), 2.89–3.02 (m, 1H), 1.95–2.11 (m, 1H), 1.80–1.94 (m, 1H), 1.40 (d, J = 6.4 Hz, 3H).
Anal. Calcd for C24H26N2O2: C 76.98; H 7.00; N 7.48. Found: C 76.83; H 7.06; N 7.46.
HPLC 99.6% (25.4 min, Inertsil ODS-3V [5 μm, 4.6 × 250 mm], 0.05% TFA in H2O/0.05% TFA in CH3CN [95:5 to 0:100/60 min]).
References
- Jump up^ Kawata, Takehisa; Tokunaga, Shin; Murai, Miki; Masuda, Nami; Haruyama, Waka; Shoukei, Youji; Hisada, Yutaka; Yanagida, Tetsuya; Miyazaki, Hiroshi; Wada, Michihito; Akizawa, Tadao; Fukagawa, Masafumi (2018). “A novel calcimimetic agent, evocalcet (MT-4580/KHK7580), suppresses the parathyroid cell function with little effect on the gastrointestinal tract or CYP isozymes in vivo and in vitro”. PLOS ONE. 13 (4): e0195316. doi:10.1371/journal.pone.0195316. PMID 29614098.
- Jump up^ Miyazaki, Hiroshi; Ikeda, Yousuke; Sakurai, Osamu; Miyake, Tsutomu; Tsubota, Rie; Okabe, Jyunko; Kuroda, Masataka; Hisada, Yutaka; Yanagida, Tetsuya; Yoneda, Hikaru; Tsukumo, Yukihito; Tokunaga, Shin; Kawata, Takehisa; Ohashi, Rikiya; Fukuda, Hajime; Kojima, Koki; Kannami, Ayako; Kifuji, Takayuki; Sato, Naoya; Idei, Akiko; Iguchi, Taku; Sakairi, Tetsuya; Moritani, Yasunori (2018). “Discovery of evocalcet, a next-generation calcium-sensing receptor agonist for the treatment of hyperparathyroidism”. Bioorganic & Medicinal Chemistry Letters. 28 (11): 2055–2060. doi:10.1016/j.bmcl.2018.04.055.
- Jump up^ “Kyowa Hakko Kirin Launches ORKEDIA® TABLETS (Evocalcet) for the Treatment of Secondary Hyperparathyroidism in Patients on Maintenance Dialysis in Japan” (Press release). Kyowa Hakko Kirin. May 22, 2018.
 |
|
| Clinical data | |
|---|---|
| Trade names | Orkedia |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| UNII | |
| Chemical and physical data | |
| Formula | C24H26N2O2 |
| Molar mass | 374.48 g·mol−1 |
///////////////Evocalcet, エボカルセト , Эвокальцет , إيفوكالسيت , 依伏卡塞 , JAPAN 2018, KHK-7580, MT-4580, UNII:E58MLH082P, ORKEDIA
SMILES Code: O=C(O)CC1=CC=C(N2C[C@@H](N[C@@H](C3=C4C=CC=CC4=CC=C3)C)CC2)C=C1