![]()
![STR1]()
PLECANATIDE; UNII-7IK8Z952OK; (3-Glutamic acid(D>E))human uroguanylin (UGN); 467426-54-6;
IUPAC Condensed
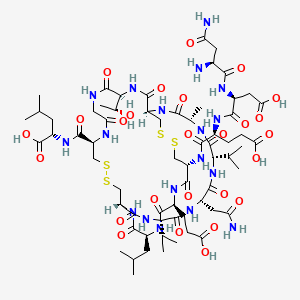
H-Asn-Asp-Glu-Cys(1)-Glu-Leu-Cys(2)-Val-Asn-Val-Ala-Cys(1)-Thr-Gly-Cys(2)-Leu-OH
(2S)-2-[[(1R,4S,7S,10S,13S,16R,21R,27S,34R,37S,40S)-10-(2-amino-2-oxoethyl)-34-[[(2S)-4-carboxy-2-[[(2S)-3-carboxy-2-[[(2S)-2,4-diamino-4-oxobutanoyl]amino]propanoyl]amino]butanoyl]amino]-37-(2-carboxyethyl)-27-[(1R)-1-hydroxyethyl]-4-methyl-40-(2-methylpropyl)-3,6,9,12,15,23,26,29,35,38,41-undecaoxo-7,13-di(propan-2-yl)-18,19,31,32-tetrathia-2,5,8,11,14,22,25,28,36,39,42-undecazabicyclo[14.13.13]dotetracontane-21-carbonyl]amino]-4-methylpentanoic acid
L-Asparaginyl-L-α-aspartyl-N-{(1R,4S,7S,10S,13S,16R,19S,22S,25R,32S,38R)-10-(2-amino-2-oxoethyl)-22-(2-carboxyethyl)-38-{[(1S)-1-carboxy-3-methylbutyl]carbamoyl}-32-[(1R)-1-hydroxyethyl]-19-isobut yl-7,13-diisopropyl-4-methyl-3,6,9,12,15,18,21,24,30,33,36-undecaoxo-27,28,40,41-tetrathia-2,5,8,11,14,17,20,23,31,34,37-undecaazabicyclo[14.13.13]dotetracont-25-yl}-L-α-glutamine
Plecanatide
RN: 467426-54-6
- Molecular FormulaC65H104N18O26S4
- Average mass1681.886 Da
-
- 105: PN: WO2012037380 SEQID: 105 claimed protein
- 1: PN: US20100152118 SEQID: 1 claimed protein
- 1: PN: WO2008151257 SEQID: 1 claimed protein
- 1: PN: WO2010065751 SEQID: 1 claimed protein
- 1: PN: WO2011069038 SEQID: 1 claimed sequence
- 1: PN: WO2012037380 SEQID: 1 claimed protein
- 1: PN: WO2014131024 SEQID: 1 claimed protein
- 1: PN: WO2015054649 SEQID: 1 claimed protein
- 20: PN: WO02078683 SEQID: 20 claimed protein
- 78: PN: WO2010065751 SEQID: 105 claimed protein
- Plecanatide
- SP 304
CLIPS
April 19, 2016
Novel Chronic Idiopathic Constipation Drug Under FDA Review
![Plecanatide is a once-daily, oral, uroguanylin analog Plecanatide is a once-daily, oral, uroguanylin analog]() Plecanatide is a once-daily, oral, uroguanylin analog
Plecanatide is a once-daily, oral, uroguanylin analogSynergy Pharmaceuticals announced the Food and Drug Administration (FDA) has accepted for review the New Drug Application (NDA) for plecanatide for the treatment of chronic idiopathic constipation (CIC).
The NDA submission was based on data from two double-blind, placebo-controlled Phase 3 trials and one open-label long term safety study in over 3,500 patients with CIC.
RELATED: NDA Submitted for Chronic Idiopathic Constipation Drug Plecanatide
The FDA has set a Prescription Drug User Fee Act (PDUFA) target action date of January 29, 2017 to make a decision on the NDA.
Plecanatide is a once-daily, oral, uroguanylin analog currently under development for the treatment of CIC and irritable bowel syndrome with constipation (IBS-C). It is designed to replicate the function of uroguanylin, a naturally occurring GI peptide, by working locally in the upper GI tract to stimulate digestive fluid movement and support regular bowel function.
PATENT
CN 104628827
http://www.google.com/patents/CN104628827A?cl=en
Prica exenatide Synergy Pharmaceuticals developed by the United States for the GC-C receptor in development of drugs, administered orally Limited.Currently underway include chronic idiopathic constipation (CIC) and constipation irritable bowel syndrome (IBS-C), including the phase III clinical trials. It is expected to receive US FDA clearance to market in recent years.Prica that peptides CAS: 467426-54-6 English name plecanatide, structural formula is as follows:
Preparation Prica that peptides from Shenzhen Han Yu medicine was first reported (CN103694320A), using a solid-phase synthesis of linear peptides in solution and then the two-step method to get into the ring, respectively. Since the method to form a ring carved in solution twice, the solution of complex composition, separation and purification difficult, the method should be improved.
Example 1
Weigh the degree of substitution of 0. 51mmol / g of Fmoc-Leu- Wang resin 10g (5. Lmmol), added to the solid phase reactor, DMF washing 3 times, the swelling 3h. The volume ratio of 1: 4 piperidine: DMF was added to the reactor the reaction, after the reaction was washed with DCM and washed twice, DMF 4 times. Weigh Fmoc-Cys (Acm) -OH 6. 34g, H0Bt 2. 07g, DIC 2. 37mL was dissolved in DMF, added to the reactor uniformly mixed, the reaction at room temperature 2h. Ninhydrin color reaction control endpoint, the resin was colorless indicates the end of the reaction, the reaction is continued if the color to colorless. After completion of the reaction, DCM was washed twice, DMF and washed 4 times.
Repeat the above steps, in accordance with the order of the sequence, followed by deprotection, coupling Fmoc-Gly-OH, Fmoc-Thr (tBu) -OH, Fmoc-Cys- (Mmt) -OH, Fmoc-Ala-OH, Fmoc- Val-OH, Fmoc-Asn (Trt) -〇H, Fmoc-Val-OH, Fmoc-Cys (Acm) -OH, Fmoc-Leu-OH, Fmoc-Glu (OtBu) -OH, Fmoc-Cys (StBu) -OH, Fmoc-Glu (OtBu) -OH, Fmoc-Asp (OtBu) -OH, Boc-Asn (Trt) -〇H〇
To a prepared peptide resin reactor volume percentage of 15% DMF solution of mercapto ethanol, reaction 2h; then DCM was added a solution of 20-fold amount DTNP reaction lh; was added after washing 1% TFA containing TIS 5% of DCM solution reaction 20min.
Preparation of peptide resin obtained after sufficiently washed with DMF, DMF was added 10 times the amount in the reaction solution 12 lh. Full wash sash.
After the preparation of the peptide resin was added in a volume ratio of 95/2/2/1 TFA / TIS / EDT / H lysis reagent 20 is added in an amount 20mL / g, the reaction ice bath lh, stirring was continued at room temperature 5h, then filtration.After lysis reagent suction filtrate using a rotary evaporator until no overflow TFA, precipitated reagent was added standing; Pulika centrifugation the precipitated crude peptide was peptide to give 8. 67g〇
The preparation of the crude peptide was obtained Pulika peptide using preparative HPLC system, wavelength 214nm, C18 reversed-phase column packing for the separation, the mobile phase of water and acetonitrile were used, with a gradient elution method to collect the target polypeptide The absorption peak. Using rotary evaporation at 30 ° C to remove most of the acetonitrile, were freeze-dried to obtain a purified Prica exenatide refined products.
Example 2
Weigh the degree of substitution of 0. 2mmol / g of Fmoc-Leu- Wang resin 10g (2mmol), added to the solid phase reactor. DMF washing 3 times, the swelling 3h. The volume ratio of 1: 4 piperidine: DMF was added to the reactor the reaction, after the reaction was washed with DCM and washed twice, DMF 4 times. Weigh Fmoc-Cys (Acm) -OH1. 24g, HOBtO. 406g, DIC 0 • 465mL dissolved in DMF solution, after mixing into the reactor at room temperature the reaction 2h.Ninhydrin color reaction control endpoint, the resin was colorless indicates the end of the reaction, the reaction is continued if the color to colorless. After completion of the reaction, DCM was washed twice, DMF and washed 4 times.
Repeat the above steps, in accordance with the order of the sequence, followed by deprotection, coupling Fmoc-Gly-OH, Fmoc-Thr (tBu) -OH, Fmoc-Cys- (Mmt) -OH, Fmoc-Ala-OH, Fmoc- Val-OH, Fmoc-Asn (Trt) -〇H, Fmoc-Val-OH, Fmoc-Cys (Acm) -OH, Fmoc-Leu-OH, Fmoc-Glu (OtBu) -OH, Fmoc-Cys (StBu) -OH, Fmoc-Glu (OtBu) -OH, Fmoc-Asp (OtBu) -OH, Boc-Asn (Trt) -〇H〇
[0053] To illustrate the preparation of the present embodiment obtained peptide resin reactor volume percent of a DMF solution of 30% mercaptoethanol, reaction 4h; then 5-fold amount DTNP in DCM reaction lh; was added after washing 1% TFA containing TIS 5% in DCM reaction 20min.
Preparation of peptide resin obtained after sufficiently washed with DMF, 20 times the amount of DMF was added in the reaction solution 12 lh. Full wash sash.
Peptide Resin [0055] Preparation was added volume ratio of 82. 5/5/5/5/2. 5 TFA / thioanisole / H20 / phenol / EDT cleavage reagents, added in an amount 10mL / g, the reaction ice bath 0 After. 5h, stirring was continued at room temperature for lh, then suction filtered. After lysis reagent suction filtrate to the non-use of force blowing TFA overflow, adding precipitation reagent standing; centrifugation precipitated Prica exenatide crude peptide to give 1. 52g.
The preparation of the crude peptide was obtained Pulika peptide using preparative HPLC system, wavelength 214nm, C18 reversed-phase column packing for the separation, the mobile phase of water and acetonitrile were used, with a gradient elution method to collect the target polypeptide The absorption peak. Using rotary evaporation at 30 ° C to remove most of the acetonitrile, were freeze-dried to obtain a purified Prica exenatide refined products.
Example 3
Weigh the degree of substitution of 0. 6mmol / g of Fmoc-Leu- Wang resin 10g (6mmol), added to the solid phase reactor, DMF washing 3 times, the swelling 3h. The volume ratio of 1: 4 piperidine: DMF was added to the reactor the reaction, after the reaction was washed with DCM and washed twice, DMF 4 times. Weigh Fmoc-Cys (Acm) -OH 7. 46g, H0Bt2. 44g, DIC 2. 79mL was dissolved in DMF, added to the reactor uniformly mixed, the reaction at room temperature 2h.Ninhydrin color reaction control endpoint, the resin was colorless indicates the end of the reaction, the reaction is continued if the color to colorless. After completion of the reaction, DCM was washed twice, DMF and washed 4 times.
Repeat the above steps, in accordance with the order of the sequence, followed by deprotection, coupling Fmoc-Gly-OH, Fmoc-Thr (tBu) -OH, Fmoc-Cys- (Mmt) -OH, Fmoc-Ala-OH, Fmoc- Val-OH, Fmoc-Asn (Trt) -〇H, Fmoc-Val-OH, Fmoc-Cys (Acm) -OH, Fmoc-Leu-OH, Fmoc-Glu (OtBu) -OH, Fmoc-Cys (StBu) -OH, Fmoc-Glu (OtBu) -OH, Fmoc-Asp (OtBu) -OH, Boc-Asn (Trt) -〇H〇
To the prepared peptide resin reactor volume percentage of 25% DMF solution of mercapto ethanol, reaction 3h; then 10-fold amount DTNP in DCM reaction lh; was added 1% TFA washed containing TIS5% DCM solution Reaction 20min〇
Preparation of peptide resin obtained after sufficiently washed with DMF, 15 times the amount of DMF was added in the reaction solution 12 lh. Full wash sash.
Preparation of the peptide resin was added in a volume ratio of 90/5/3/2 TFA / thioanisole / anisole / EDT cleavage reagents, added in an amount 20mL / g, the ice bath was reacted 0.lh, stirring was continued at room temperature The reaction 10h, then filtration. After lysis reagent suction filtrate using a rotary evaporator until no overflow TFA, precipitated reagent was added standing; Pulika centrifugation the precipitated crude peptide was peptide to give 8. 46g.
The preparation of the crude peptide was obtained Pulika peptide using preparative HPLC system, wavelength 214nm, C18 reversed-phase column packing for the separation, the mobile phase of water and acetonitrile were used, with a gradient elution method to collect the target polypeptide The absorption peak. Using rotary evaporation at 30 ° C to remove most of the acetonitrile, were freeze-dried to obtain a purified Prica exenatide refined products.
Although the above has been described with general, specific embodiments and test, the present invention has been described in detail, but on the basis of the present invention, it may make some changes or improvements, which the skilled artisan It is obvious. Thus, the present invention without departing from the spirit on the basis of these modifications or improvements made, belong to the scope of the invention as claimed.
PATENT
CN 104211777
http://www.google.com/patents/CN104211777A?cl=en
The pickup exenatide (Plecanatide) is a synthetic analogue of guanylin urine (urine guanylin is a natriuretic hormone, can regulate gastrointestinal transport of ions and liquid), pickup exenatide enter After in vivo and guanylate gastrointestinal tract endothelial cells cyclase C binding and activation, activation of the cystic fibrosis transmembrane conductance regulator (CFTR), to promote chloride and water into the intestine, thereby promoting bowel motility, improve constipation symptoms.
Synergy company announced its pick in the research of new drugs that peptide (code: SP304) on October 6, 2010 the treatment of gastrointestinal disorders II a clinical experimental results. The study, conducted in patients with chronic constipation showed that the drugs can improve bowel function in patients, promote intestinal motility and reduce abdominal discomfort shape. In the experiment, there was no diarrhea and other adverse reactions, at the doses tested did not detect the pickup system that peptides are absorbed. The drug is expected for the treatment of chronic constipation (CC), constipation-predominant irritable bowel syndrome (IBS-C) and other gastrointestinal disorders. CC and IBS-C is a common gastrointestinal disease that can cause serious impact on the work and the quality of life of patients. Synergy will continue to conduct clinical trials of other pickups that peptide.
The structure of the peptide pickup that is:
H-Asn-Asp-Asp-Cys-Glu-Leu-Cys-Val-Asn-Val-Ala-Cys-Thr-Gly-C ys-Leu-〇H (4-12 disulfide, 7- 15)
Example 30:
H-Asn-Asp-Asp-Cys-Glu-Leu-Cys-Val-Asn-Val-Ala-Cys-Thr-Gly-C ys-Leu-〇H (4-12 disulfide, 7- 15) Preparation of
embodiments will be prepared by the method of Example 18 H-Asn (Trt) -Asp (OtBu) -Asp (OtBu) -Cys (mmt) -Glu (Ot Bu) -Leu-Cys (StBu) -Val-Asn ( Trt) -Val-Ala-Cys (mmt) -Thr (tBu) -Gly-Cys (StBu) -Leu-CT C resin (IOOmmol, 472. 88g) disposed cracking reactor to 10ml / g resin ratio Add lysis reagent (TFA: EDT: water = 95: 2 5:.. 2 5 (V / V)), stirred at room temperature 2h. The reaction was filtered with sand core funnel, and then added a small amount of TFA The resin was washed in the funnel, collecting the filtrate, the combined filtrate was concentrated. Frozen in dry diethyl ether was added (100ml / g peptide purpose tree months) and the solution was precipitated, centrifuged to remove the precipitate was washed with diethyl ether after dry ether three times, and dried in vacuo to give a white solid powder was approximately 180g, i.e., H-Asn-Asp-Asp -Cys-Glu-Leu-Cys (StBu) -Val-Asn-Val-Ala-Cys-Thr-Gly-Cy s (StBu) -Leu-OH. The solid was dissolved with water to lmg / ml solution. Was added an aqueous solution of 1% by volume of H2O2, the reaction was stirred at room temperature 30min, to prepare H-Asn-Asp-Asp-Cys-Glu-Leu-Cys (StBu) -Val-Asn-Val-Ala-Cys-Thr-Gl y-Cys (StBu) -Leu-OH (disulfide 4-12) was treated with a rotary evaporator after drying the compound containing 500ml 20% β- mercaptoethanol and 0. IM N- methylmorpholine were dissolved in water, followed by stirring After 12h the reaction, the reaction solution was diluted with water to 3mg / ml was about 60L, dissolved in ethanol was added with IL 300mmol I2 solution, the reaction was stirred at room temperature 2h.Adding an appropriate amount Vc remove excess I2, until the color of the reaction solution was transparent, i.e., to give H-Asn-Asp-Asp-Cys-Glu-Leu-Cys-Val-As n-Val-Ala-Cys-Thr-Gly-Cys-L eu_0H (disulfide bonds 4-12, 7-15).
PATENT
WO 2014197720
CN 103694320
WO 2012118972
WO 2012037380
WO 2011069038
US 20100152118
WO 2010065751
///Plecanatide, 普卡那肽 , ليكاناتيد , плеканатид, 467426-54-6, Chronic Idiopathic Constipation, NDA, SP 304, SYNERGY, PEPTIDE,
C[C@H]1C(=O)N[C@H]2CSSC[C@@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)N[C@@H](CSSC[C@H](NC(=O)CNC(=O)[C@@H](NC2=O)[C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)O)C(=O)N[C@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)N1)C(C)C)CC(=O)N)C(C)C)CC(C)C)CCC(=O)O)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CC(=O)N)N
OR
O=C(N[C@@H](CC(=O)O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@H]1CSSC[C@@H]2NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CCC(=O)O)NC1=O)CC(C)C)CSSC[C@H](NC(=O)CNC(=O)[C@@H](NC2=O)[C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)O)C(C)C)C(C)C)[C@@H](N)CC(N)=O