FDA approves Intrarosa for postmenopausal women experiencing pain during sex
The U.S. Food and Drug Administration approved Intrarosa (prasterone) to treat women experiencing moderate to severe pain during sexual intercourse (dyspareunia), a symptom of vulvar and vaginal atrophy (VVA), due to menopause. Intrarosa is the first FDA approved product containing the active ingredient prasterone, which is also known as dehydroepiandrosterone (DHEA).
For Immediate Release
November 17, 2016
Release
The U.S. Food and Drug Administration approved Intrarosa (prasterone) to treat women experiencing moderate to severe pain during sexual intercourse (dyspareunia), a symptom of vulvar and vaginal atrophy (VVA), due to menopause. Intrarosa is the first FDA approved product containing the active ingredient prasterone, which is also known as dehydroepiandrosterone (DHEA).
During menopause, levels of estrogen decline in vaginal tissues, which may cause a condition known as VVA, leading to symptoms such as pain during sexual intercourse.
“Pain during sexual intercourse is one of the most frequent symptoms of VVA reported by postmenopausal women,” said Audrey Gassman, M.D., deputy director of the Division of Bone, Reproductive, and Urologic Products (DBRUP) in the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research (CDER). “Intrarosa provides an additional treatment option for women seeking relief of dyspareunia caused by VVA.”
Efficacy of Intrarosa, a once-daily vaginal insert, was established in two 12-week placebo-controlled clinical trials of 406 healthy postmenopausal women, 40 to 80 years of age, who identified moderate to severe pain during sexual intercourse as their most bothersome symptom of VVA. Women were randomly assigned to receive Intrarosa or a placebo vaginal insert. Intrarosa, when compared to placebo, was shown to reduce the severity of pain experienced during sexual intercourse.
The safety of Intrarosa was established in four 12-week placebo-controlled trials and one 52-week open-label trial. The most common adverse reactions were vaginal discharge and abnormal Pap smear.
Although DHEA is included in some dietary supplements, the efficacy and safety of those products have not been established for diagnosing, curing, mitigating, treating or preventing any disease.
Intrarosa is marketed by Quebec-based Endoceutics Inc.
The FDA, an agency within the U.S. Department of Health and Human Services, protects the public health by assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices. The agency also is responsible for the safety and security of our nation’s food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products.
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
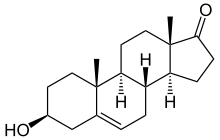
(3S,8R,9S,10R,13S,14S)-3-hydroxy-10,13-dimethyl-1,2,3,4,7,8,9,11,12,14,15,16-dodecahydrocyclopenta[a]phenanthren-17-one; (1S,2R,5S,10R,11S,15S)-5-Hydroxy-2,15-dimethyltetracyclo[8.7.0.02,7.011,15]heptadec-7-en-14-one
|
|
| Clinical data | |
| Routes of administration |
Oral |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Biological half-life | 12 hours |
| Excretion | Urinary:?% |
| Identifiers | |
| CAS Number | 53-43-0  |
| ATC code | A14AA07 (WHO) G03EA03 (WHO) (combination with estrogen) |
| PubChem | CID 5881 |
| IUPHAR/BPS | 2370 |
| DrugBank | DB01708  |
| ChemSpider | 5670  |
| UNII | 459AG36T1B  |
| ChEBI | CHEBI:28689  |
| ChEMBL | CHEMBL90593  |
| Synonyms | (3β)-3-Hydroxyandrost-5-en-17-one |
| Chemical data | |
| Formula | C19H28O2 |
| Molar mass | 288.424 g/mol |
| 3D model (Jmol) | Interactive image |
Page Last Updated: 11/17/2016
Note: If you need help accessing information in different file formats, see Ins
Filed under: FDA 2016 Tagged: (DHEA), Approves, dehydroepiandrosterone, fda, Intrarosa, pain during sex, postmenopausal women, prasterone
![]()





















