| Republic of Uzbekistan Oʻzbekiston Respublikasi (Uzbek) |
|---|
| FlagState emblem |
ZF2001
ZIFIVAX
CAS 2609662-31-7
A COVID-19 vaccine comprising a dimeric form of SARS-CoV-2 receptor-binding domain (RBD) produced in China hamster ovary (CHO) cells and adjuvanted with aluminum hydroxide (Anhui Zhifei Longcom/Institute of Microbiol. China Academy of Sciences)
Recombinant vaccine
Anhui Zhifei Longcom Biopharmaceutical, Institute of Microbiology of the Chinese Academy of Sciences
China, Uzbekistan
CHO Cells Recombinant Vaccine
- ZF-2001
- ZF-UZ-VAC2001
- Chinese Academy of Sciences (Originator)
- Zhifei Longcom (Originator)
Human SARS-CoV-2 (Covid-19 coronavirus) vaccine consisting of recombinant dimer comprising two RBD domains (R319-K527) of the spike glycoprotein of SARS-CoV-2 fused via a disulfide link; expressed in CHO cells
ZF-2001 is a recombinant coronavirus vaccine jointly developed by the Institute of Microbiology of the Chinese Academy of Sciences and Zhifei Longcom. The vaccine became available in 2021 in Uzbekistan under an emergency use authorization for the prevention of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (COVID-19). The vaccine is currently evaluated in phase III clinical trials.
This vaccine candidate, developed in China, uses SARS-CoV-2 protein subunits that are entirely engineered, created, and secreted by Chinese Hamster Ovary (CHO) cells1. The vaccine candidate is sponsored by Anhui Zhifei Longcom Biologic Pharmacy Co., Ltd. and is undergoing phase I clinical trials to evaluate safety and tolerability.
ZF2001, trade-named ZIFIVAX, is an adjuvanted protein subunit COVID-19 vaccine developed by Anhui Zhifei Longcom in collaboration with the Institute of Microbiology at the Chinese Academy of Sciences.[1][2] As of December 2020, the vaccine candidate was in Phase III trials with 29,000 participants in China, Ecuador, Malaysia, Pakistan, and Uzbekistan.[3][4]
ZF2001 employs technology similar to other protein-based vaccines in Phase III trials from Novavax, Vector Institute, and Medicago.[5] It is administered in 3 doses over a period of 2 months.[6]
ZF2001 was first approved for use in Uzbekistan and later China.[7][8] Production capacity is expected to be one billion doses a year.[6] Phase II results published in The Lancet on the three dose administration showed seroconversion rates of neutralizing antibodies of between 92% to 97%.[9]
Anhui Zhifei Longcom Biopharmaceuticals began a phase 3 clinical trial for its recombinant protein vaccine candidate in December, according to the WHO. State-run China Global Television Network in November reported that a one-year trial would take place in Uzbekistan and aim to recruit 5,000 volunteers. Anhui Zhifei is a unit of private firm Chongqing Zhifei Biological Products. It is co-developing the vaccine with the Chinese Academy of Sciences, a government institution.
Emergency Use Authorization received in UZ by Zhifei Longcom for the prevention of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (COVID-19)
Description
As described in Cell, the CoV spike receptor-binding domain (RBD) is an attractive vaccine target for coronaviruses but is constrained by limited immunogenicity, however a dimeric form of MERS-CoV RBD offers greater protection. The RBD-dimer significantly increases neutralizing antibodies compared to a conventional monomeric form and protected mice against MERS-CoV infection. CoV RBD-dimer have been produced at high yields in pilot scale production.[10]
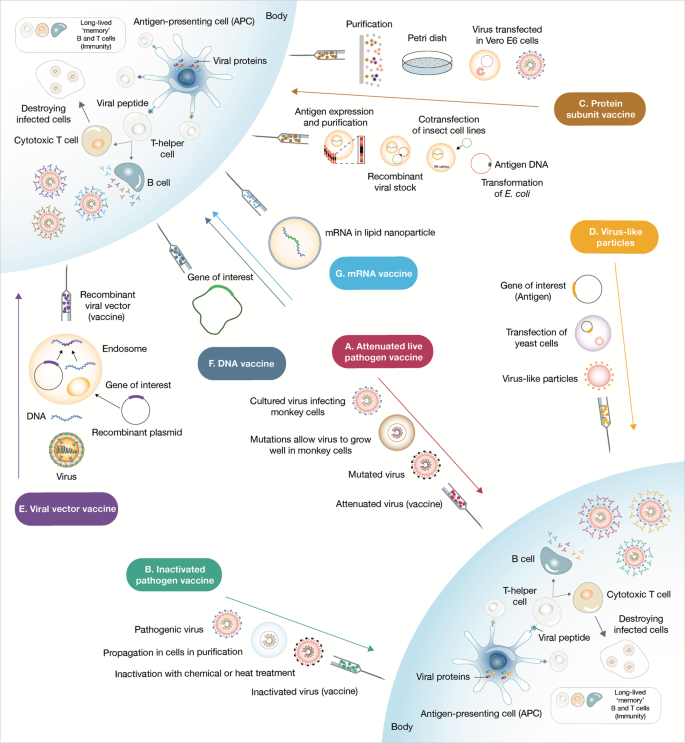
Rather than injecting a whole virus, subunit vaccines contains virus particles specially selected to stimulate an immune response. Because the fragments are incapable of causing disease, subunit vaccines are considered very safe.[11] Subunit vaccines in widespread use include the Hepatitis B vaccine and Pertussis vaccine. However, as only a few viral components are included in the vaccine which does not display the full complexity of the virus, their efficacy may be limited.[12] Subunit vaccines are delivered alongside adjuvants and booster doses may be required.[11]
According to industry experts, production for this kind of vaccine is stable and reliable, and easier to achieve large-scale industrial production at home and overseas. However it was noted it can be very inconvenient for people to come back for a second and third dose.[6]
ZF2001 (Anhui Zhifei Longcom Biopharmaceutical/Chinese Academy of Medical Sciences)
The latest subunit vaccine candidate to enter Phase 3 clinical studies is the adjuvanted RBD-dimeric antigen designed by Anhui Zhifei Longcom Biopharmaceutical and the Institute of Microbiology of the Chinese Academy of Medical Sciences. Phase 3 clinical study was launched on December104 and will be initially carried out in China and Uzbekistan while Indonesia, Pakistan and Ecuador will follow as study sites (Clinical Trial Identifier: NCT04646590 and Registration Number: ChiCTR2000040153). The design of the study involves recruitment of 22,000 volunteers from China and 7000 subjects outside China for a total of 29,000 volunteers. There are still no published results on this candidate, however data from its Phase 2 placebo-controlled clinical trial (Clinical Trial Identifier: NCT04466085) conducted on a total of 900 participants ranging from 18 to 59 years old suggest that a 2 or 3 dose regimen is evaluated. Each immunization will be separated by the next by 4 weeks.
Development
Phase I and II trials and results
In June, Longcom began a double-blind, randomized, placebo parallel controlled Phase I trial with 50 participants aged 18–59 in Chongqing divided into low-dose, high-dose, and placebo groups.[13]
In July, Longcom began a randomized, double-blind, placebo-controlled Phase II trial with 900 participants aged 18–59 in Changsha, Hunan divided into low-dose, high-dose, and placebo groups.[14] In August, an additional Phase II trial was launched with 50 participants aged 60 and above.[15][1]
In Phase II results published in The Lancet, on the two-dose schedule, seroconversion rates of neutralizing antibodies after the second dose were 76% (114 of 150 participants) in a 25 μg group and 72% (108 of 150) in a 50 μg group. On the three-dose schedule, seroconversion rate of neutralizing antibodies after the third dose were 97% (143 of 148 participants) in the 25 μg group and 93% (138 of 148) in the 50 μg group. 7 to 14 days after the administration of the third dose, the GMTs of neutralizing antibodies reached levels that were significantly higher than observed in human convalescent serum of recovering COVID-19 patients, especially in the 25 μg group.[9]
Phase III trials
In December, Longcom began enrollment of a Phase III randomized, double-blind, placebo-controlled clinical trial for 29,000 participants, including 750 participants between 18-59 and 250 participants 60 and older in China and 21,000 participants between 18-59 and 7,000 participants 60 and older outside China.[16][17]
In December, Malaysia‘s MyEG announced it would conduct Phase III trials. If the trials were successful, MyEG would be the sole distributor of ZF2001 in Malaysia for 3 years.[4]
In December, Uzbekistan began a year-long Phase III trial of ZF2001 with 5,000 volunteers between 18 and 59.[18][19]
In December, Ecuador‘s Minister of Health, Juan Carlos Zevallos announced Phase III trials would involve between 5,000 and 8,000 volunteers.[20]
In February, Pakistan‘s Drug Regulatory Authority (DRAP) approved Phase III trials with approximately 10,000 participants to be conducted at UHS Lahore, National Defense Hospital, and Agha Khan Hospital.[21]
Discussions to begin Phase III trials are also underway in Indonesia.[17][22]
COVID-19 Variants
In February, lab studies of twelve serum samples taken from recipients of BBIBP-CorV and ZF2001 retained neutralizing activity against the Beta variant although with weaker activity than against the original virus.[23] For ZF-2001, geometric mean titers declined by 1.6-fold, from 106.1 to 66.6, which was less than antisera from mRNA vaccine recipients with a 6-folds decrease.[24] Preliminary clinical data from Novavax and Johnson & Johnson also showed they were less effective in preventing COVID-19 in South Africa, where the new variant is widespread.[23]
Manufacturing
The company’s vaccine manufacturing facility was put into use in September.[17] In February 2021, Pu Jiang, General Manager of Zhifei Longcom, said the company had an annual production capacity of 1 billion doses.[6]
Marketing and deployment
| Full authorization Emergency authorization |
See also: List of COVID-19 vaccine authorizations § RBD-Dimer
On March 1, Uzbekistan granted approval for ZF2001 (under tradename ZF-UZ-VAC 2001) after having taken part in the Phase III trials.[8] In March, Uzbekistan received 1 million doses and started vaccinations in April.[25] By May, a total of 3 million doses had been delivered.[26]
On March 15, China approve of ZF2001 for emergency use after being approved by Uzbekistan earlier in the month.[7]
References
- ^ Jump up to:a b “Anhui Zhifei Longcom: RBD-Dimer – COVID19 Vaccine Tracker”. covid19.trackvaccines.org. Retrieved 27 December2020.
- ^ “COVID-19 Vaccine: ZIFIVAX by Anhui Zhifei Longcom Biopharma, Institute of Microbiology Chinese Academy of Sciences”. covidvax.org. Retrieved 27 December 2020.
- ^ “Fifth Chinese Covid-19 vaccine candidate ready to enter phase 3 trials”. South China Morning Post. 20 November 2020. Retrieved 27 December 2020.
- ^ Jump up to:a b Ying TP (7 December 2020). “MYEG to conduct phase 3 clinical trial for China’s Covid-19 vaccine in Msia | New Straits Times”. NST Online. Retrieved 27 December 2020.
- ^ Zimmer C, Corum J, Wee SL (10 June 2020). “Coronavirus Vaccine Tracker”. The New York Times. ISSN 0362-4331. Retrieved 27 December 2020.
- ^ Jump up to:a b c d “China’s production bottleneck ‘could be eased with latest Covid-19 vaccine'”. South China Morning Post. 17 March 2021. Retrieved 18 March 2021.
- ^ Jump up to:a b Liu, Roxanne (15 March 2021). “China IMCAS’s COVID-19 vaccine obtained emergency use approval in China”. Reuters. Retrieved 15 March 2021.
- ^ Jump up to:a b Mamatkulov, Mukhammadsharif (1 March 2021). “Uzbekistan approves Chinese-developed COVID-19 vaccine”. Reuters. Retrieved 2 March 2021.
- ^ Jump up to:a b Yang, Shilong; Li, Yan; Dai, Lianpan; Wang, Jianfeng; He, Peng; Li, Changgui; Fang, Xin; Wang, Chenfei; Zhao, Xiang; Huang, Enqi; Wu, Changwei (24 March 2021). “Safety and immunogenicity of a recombinant tandem-repeat dimeric RBD-based protein subunit vaccine (ZF2001) against COVID-19 in adults: two randomised, double-blind, placebo-controlled, phase 1 and 2 trials”. The Lancet Infectious Diseases. 0. doi:10.1016/S1473-3099(21)00127-4. ISSN 1473-3099. PMC 7990482. PMID 33773111.
- ^ Dai L, Zheng T, Xu K, Han Y, Xu L, Huang E, et al. (August 2020). “A Universal Design of Betacoronavirus Vaccines against COVID-19, MERS, and SARS”. Cell. 182 (3): 722–733.e11. doi:10.1016/j.cell.2020.06.035. PMC 7321023. PMID 32645327.
- ^ Jump up to:a b “What are protein subunit vaccines and how could they be used against COVID-19?”. http://www.gavi.org. Retrieved 27 December2020.
- ^ Dong Y, Dai T, Wei Y, Zhang L, Zheng M, Zhou F (October 2020). “A systematic review of SARS-CoV-2 vaccine candidates”. Signal Transduction and Targeted Therapy. 5 (1): 237. doi:10.1038/s41392-020-00352-y. PMC 7551521. PMID 33051445.
- ^ Clinical trial number NCT04445194 for “Phase I Clinical Study of Recombinant Novel Coronavirus Vaccine” at ClinicalTrials.gov
- ^ Clinical trial number NCT04466085 for “A Randomized, Blinded, Placebo-controlled Trial to Evaluate the Immunogenicity and Safety of a Recombinant New Coronavirus Vaccine (CHO Cell) With Different Doses and Different Immunization Procedures in Healthy People Aged 18 to 59 Years” at ClinicalTrials.gov
- ^ Clinical trial number NCT04550351 for “A Randomized, Double-blind, Placebo-controlled Phase I Clinical Trial to Evaluate the Safety and Tolerability of Recombinant New Coronavirus Vaccines (CHO Cells) in Healthy People Aged 60 Years and Above” at ClinicalTrials.gov
- ^ Clinical trial number NCT04646590 for “A Phase III Randomized, Double-blind, Placebo-controlled Clinical Trial in 18 Years of Age and Above to Determine the Safety and Efficacy of ZF2001, a Recombinant Novel Coronavirus Vaccine (CHO Cell) for Prevention of COVID-19” at ClinicalTrials.gov
- ^ Jump up to:a b c “Another Chinese Covid-19 vaccine enters late-stage human trials with a plan to produce 300 million doses annually”. Business Insider. Retrieved 27 December 2020.
- ^ Reuters Staff (11 November 2020). “Uzbekistan to carry out late-stage trial of Chinese COVID-19 vaccine candidate”. Reuters. Retrieved 27 December 2020.
- ^ “Uzbekistan poised to start trials on Chinese COVID-19 vaccine | Eurasianet”. eurasianet.org. Retrieved 27 December 2020.
- ^ “Ecuador participará en ensayos de una vacuna china contra el covid-19”. CNN (in Spanish). 29 December 2020. Retrieved 23 January 2021.
- ^ “China’s third vaccine enters Pakistan”. The Nation. 15 February 2021. Retrieved 28 February 2021.
- ^ “Covid vaccine tracker: How do the leading jabs compare?”. http://www.ft.com. 23 December 2020. Retrieved 27 December 2020.
- ^ Jump up to:a b Liu, Roxanne (3 February 2021). “Sinopharm’s COVID-19 vaccine remained active against S.Africa variant, effect reduced – lab study”. Reuters. Retrieved 29 March 2021.
- ^ Huang, Baoying; Dai, Lianpan; Wang, Hui; Hu, Zhongyu; Yang, Xiaoming; Tan, Wenjie; Gao, George F. (2 February 2021). “Neutralization of SARS-CoV-2 VOC 501Y.V2 by human antisera elicited by both inactivated BBIBP-CorV and recombinant dimeric RBD ZF2001 vaccines”. bioRxiv: 2021.02.01.429069. doi:10.1101/2021.02.01.429069.
- ^ uz, Kun. “Uzbekistan receives 1 million doses of ZF-UZ-VAC 2001 vaccine”. Kun.uz. Retrieved 28 March 2021.
- ^ Romakayeva, Klavdiya (18 May 2021). “Uzbekistan receives third batch of Chinese-Uzbek COVID-19 vaccine”. Trend.Az. Retrieved 19 May 2021.
| Vaccine description | |
|---|---|
| Target | SARS-CoV-2 |
| Vaccine type | Protein subunit |
| Clinical data | |
| Trade names | ZIFIVAX |
| Routes of administration | Intramuscular |
| ATC code | None |
| Identifiers | |
| DrugBank | DB15893 |
| Part of a series on the |
| COVID-19 pandemic |
|---|
| COVID-19 (disease)SARS-CoV-2 (virus) |
| showTimeline |
| showLocations |
| showInternational response |
| showMedical response |
| showImpact |
| COVID-19 portal |
////////ZF2001, ZIFIVAX, corona virus, covid 19, SARS-CoV-2, ZF 2001, ZF-UZ-VAC2001, Uzbekistan, approvals 2021
